Journal Description
ChemEngineering
ChemEngineering
is an international, peer-reviewed, open access journal on the science and technology of chemical engineering, published bimonthly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, ESCI (Web of Science), Inspec, CAPlus / SciFinder, and other databases.
- Journal Rank: CiteScore - Q1 (General Engineering)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 17.2 days after submission; acceptance to publication is undertaken in 6.8 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
2.5 (2022);
5-Year Impact Factor:
2.7 (2022)
Latest Articles
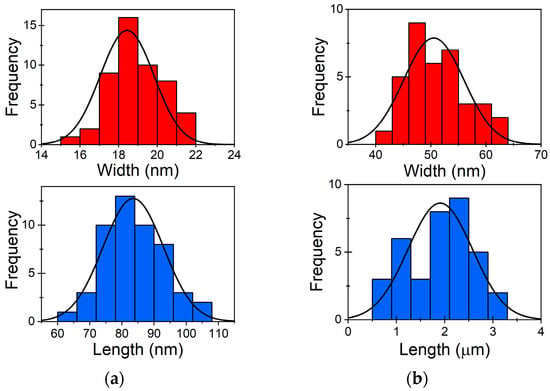
Heat-Induced Fragmentation and Adhesive Behaviour of Gold Nanowires for Surface-Enhanced Raman Scattering Substrates
ChemEngineering 2024, 8(1), 15; https://doi.org/10.3390/chemengineering8010015 - 09 Jan 2024
Abstract
This study explores a novel approach to surface-enhanced Raman scattering (SERS) substrate fabrication through the heat-induced fragmentation of gold nanowires (Au NWs) and its impact on gold nanoparticle adhesion/static friction using atomic force microscopy manipulations. Controlled heating experiments and scanning electron microscopy measurements
[...] Read more.
This study explores a novel approach to surface-enhanced Raman scattering (SERS) substrate fabrication through the heat-induced fragmentation of gold nanowires (Au NWs) and its impact on gold nanoparticle adhesion/static friction using atomic force microscopy manipulations. Controlled heating experiments and scanning electron microscopy measurements reveal significant structural transformations, with NWs transitioning into nanospheres or nanorods in a patterned fashion at elevated temperatures. These morphological changes lead to enhanced Raman signals, particularly demonstrated in the case of Rhodamine B molecules. The results underscore the critical role of NW shape modifications in augmenting the SERS effect, shedding light on a cost-effective and reliable method for producing SERS substrates.
Full article
(This article belongs to the Topic Advances in Chemistry and Chemical Engineering)
►
Show Figures
Open AccessArticle
Oxidation of Allura Red AC Using the NaHCO3-activated H2O2 System Catalyzed with Cobalt Supported on Al-PILC
by
, , , , and
ChemEngineering 2024, 8(1), 14; https://doi.org/10.3390/chemengineering8010014 - 08 Jan 2024
Abstract
The oxidation of aqueous solutions containing Allura Red AC (AR–AC) using bicarbonate-activated peroxide (BAP) and cobalt-impregnated pillared clay (Co/Al–PILC) as the catalyst was investigated. Using the CCD-RMS approach (central composite design–response surface methodology), the effects of dye, H2O2, and
[...] Read more.
The oxidation of aqueous solutions containing Allura Red AC (AR–AC) using bicarbonate-activated peroxide (BAP) and cobalt-impregnated pillared clay (Co/Al–PILC) as the catalyst was investigated. Using the CCD-RMS approach (central composite design–response surface methodology), the effects of dye, H2O2, and NaHCO3 concentrations on AR–AC degradation were studied. The decolorization, total nitrogen (TN), and total carbon (TC) removals were the analyzed responses, and the experimental data were fitted to empirical quadratic equations for these responses, obtaining coefficients of determination R2 and adjusted-R2 higher than 0.9528. The multi-objective optimization conditions were [dye] = 21.25 mg/L, [H2O2] = 2.59 mM, [NaHCO3] = 1.25 mM, and a catalyst loading of 2 g/L. Under these conditions, a decolorization greater than 99.43% was obtained, as well as TN and TC removals of 72.82 and 18.74%, respectively, with the added advantage of showing cobalt leaching below 0.01 mg/L. Chromatographic analyses (GC–MS and HPLC) were used to identify some reaction intermediates and by-products. This research showed that wastewater containing azo dyes may be treated using the cobalt-catalyzed BAP system in heterogeneous media.
Full article
(This article belongs to the Topic Advances in Chemistry and Chemical Engineering)
►▼
Show Figures
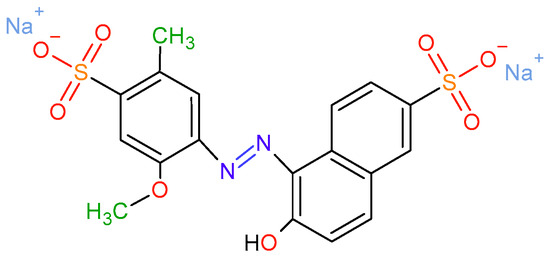
Figure 1
Open AccessArticle
Evaluation of Ceramic Properties of Bauxitic Materials from SE of Iberian Range
by
, , , , , , and
ChemEngineering 2024, 8(1), 13; https://doi.org/10.3390/chemengineering8010013 - 08 Jan 2024
Abstract
►▼
Show Figures
The use of aluminum-rich clays and bauxites as refractory materials is common. Upon firing, these materials form mullite crystals in the shape of needles embedded in a siliceous and vitreous matrix, with mullite being responsible for the refractory properties. In this study, bauxite
[...] Read more.
The use of aluminum-rich clays and bauxites as refractory materials is common. Upon firing, these materials form mullite crystals in the shape of needles embedded in a siliceous and vitreous matrix, with mullite being responsible for the refractory properties. In this study, bauxite samples for use in refractory applications have been characterized. Chemical analysis revealed that the alumina content varied between 34 and 40%, with silica values generally being high (around 40%), except for one sample (26%). Two samples were found to be the most suitable for use as “refractory clay” refractories. However, high silica or Fe oxide contents can affect mineralogical transformations at high temperatures. Mineralogical analysis confirmed the presence of several minerals in the bauxite materials, including kaolinite, halloysite, anatase, rutile, gibbsite and boehmite. Differential thermal analysis (DTA) showed the decomposition of gibbsite and its partial transformation to boehmite and alumina, and the dehydroxylation of kaolinite, with primary mullite crystallization observed at a high temperature. These findings provide valuable information for the selection and optimization of bauxite materials for refractory applications, considering their chemical composition and mineralogical characteristics.
Full article
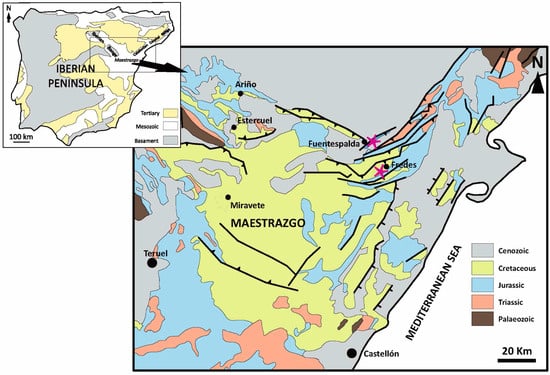
Figure 1
Open AccessArticle
Different Substrate Selectivity and Product Patterns of Immobilized Thermophilic Lipases from Geobacillus stearothermophilus, Anoxybacillus flavithermus, and Thermomyces lanuginosus for Glyceryl Decanoate Synthesis
ChemEngineering 2024, 8(1), 12; https://doi.org/10.3390/chemengineering8010012 - 05 Jan 2024
Abstract
►▼
Show Figures
Lipases can catalyze synthesis reactions in a micro aqueous system, producing useful partial glycerides (mono- and diglycerides), and these compounds are commonly utilized in different products as surfactants. Depending on the microbial sources for lipases, immobilization conditions, and starting substrates for synthesis reaction,
[...] Read more.
Lipases can catalyze synthesis reactions in a micro aqueous system, producing useful partial glycerides (mono- and diglycerides), and these compounds are commonly utilized in different products as surfactants. Depending on the microbial sources for lipases, immobilization conditions, and starting substrates for synthesis reaction, the composition and yields of the resulting partial glycerides could be variable. These differences could lead to the final efficacy of partial glycerides as surfactants in targeted products. Therefore, it is necessary to establish a group of immobilized lipases from different microbial sources with information about substrate specificity to produce effective partial glycerides for various product types. Here, lipases from thermophilic Geobacillus stearothermophilus and Anoxybacillus flavithermus were prepared with a simple partial purification method, and after immobilization, these lipases were tested to synthesize partial glycerides using different types of decanoic acids. The distinct product patterns were analyzed using HPLC. Both immobilized lipases showed the highest substrate selectivity to decanoic acids in common, producing mainly glyceryl monodecanoate. However, commercial immobilized lipases from Thermomyces lanuginosus produced the largest glyceryl monodecanoate from methyl decanoate. These results indicate the importance of immobilization conditions like different microbial sources and substrates and the need for their optimal combination.
Full article
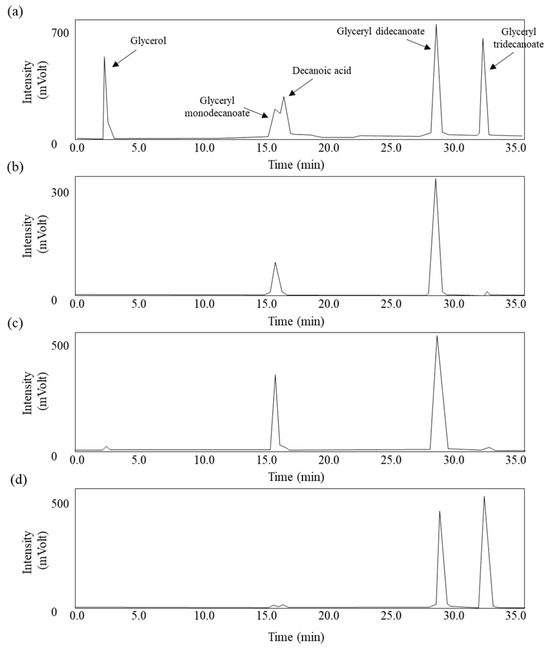
Figure 1
Open AccessArticle
Sequential Extraction of Carbohydrates and Lipids from Chlorella vulgaris Using Combined Physical and Chemical Pre-Treatments
ChemEngineering 2024, 8(1), 11; https://doi.org/10.3390/chemengineering8010011 - 05 Jan 2024
Abstract
A key focus of microalgae-based fuels/chemicals research and development has been on the lipids that many strains generate, but recent studies show that solely recovering these lipids may not be cost competitive with fossil-derived processes. However, if the carbohydrates can also be recovered
[...] Read more.
A key focus of microalgae-based fuels/chemicals research and development has been on the lipids that many strains generate, but recent studies show that solely recovering these lipids may not be cost competitive with fossil-derived processes. However, if the carbohydrates can also be recovered and ultimately converted into useful chemical intermediates, this may improve the economics for microalgae-based sustainable product technologies. In the present work, physical and chemical pre-treatments were performed on the Chlorella vulgaris microalgae strain to recover the carbohydrates from the biomass primarily in the form of glucose and galactose. The effects of temperature, acid concentration, microalgae solid-to-liquid loading, and hydrolysis time on carbohydrate hydrolysis and recovery was explored to identify optimum conditions. The highest recovery of total carbohydrates, 90 ± 1.1 wt% at 95% confidence which represents 40 wt% of the initial biomass, was obtained using temperature-assisted weak-acid extraction. Sequential extraction of carbohydrates and lipids was then explored. The highest recovery of total lipids was 71 ± 1.8 wt%, which represents 22 ± 0.9 wt% of the initial biomass. The sequential extraction of carbohydrates followed by lipids resulted in an overall recovery of 60 ± 1.6 wt% of the initial biomass, which is higher than current single product recovery strategies. These results suggest that adding carbohydrate recovery may be a viable strategy for overcoming a major economic hurdle to microalgae-derived chemical and fuel production by significantly increasing the yield of usable materials from microalgae biomass.
Full article
(This article belongs to the Special Issue Process Intensification for Chemical Engineering and Processing)
►▼
Show Figures

Figure 1
Open AccessArticle
Simulation Process for Allyl Alcohol Production via Deoxydehydration of Glycerol
ChemEngineering 2024, 8(1), 10; https://doi.org/10.3390/chemengineering8010010 - 03 Jan 2024
Abstract
A process for the deoxydehydration (DODH) of glycerol to allyl alcohol in 2-hexanol as solvent was modelled with Aspen Plus. Experimental results for the DODH reaction, the liquid vapour equilibria and the catalytic hydrogenation were employed for the development of the model. The
[...] Read more.
A process for the deoxydehydration (DODH) of glycerol to allyl alcohol in 2-hexanol as solvent was modelled with Aspen Plus. Experimental results for the DODH reaction, the liquid vapour equilibria and the catalytic hydrogenation were employed for the development of the model. The whole process consists of four subsystems: allyl alcohol production (S1), solvent recovery (S2), allyl alcohol purification (S3) and solvent regeneration (S4). Based on the results of the process model, allyl alcohol with 96% yield and a purity of 99.99% with product loss of only 0.2% was obtained. The optimisation of the energy consumption through an integrated heat exchange network resulted in a net primary energy input of 863.5 kW, which corresponded to a carbon footprint of 1.89 kgCO2/kgAllylOH.
Full article
(This article belongs to the Collection Green and Environmentally Sustainable Chemical Processes)
►▼
Show Figures
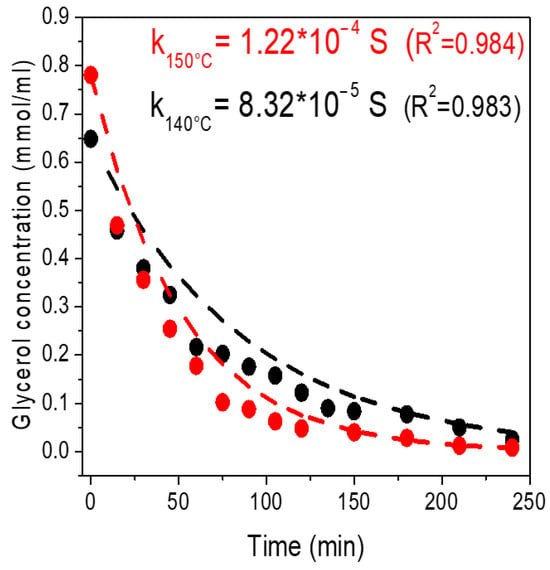
Figure 1
Open AccessArticle
Nano-Iron Oxide Coating for Enhanced Heat Transfer in Gas–Solid Fluidized Bed Systems
by
, , , , and
ChemEngineering 2024, 8(1), 9; https://doi.org/10.3390/chemengineering8010009 - 02 Jan 2024
Abstract
►▼
Show Figures
This study explores using iron oxide coatings on glass beads to improve heat transfer efficiency in fluidized bed reactors. Techniques such as BET surface area analysis, SEM imaging, and X-ray diffraction were used to characterize the coated beads. Results showed the successful creation
[...] Read more.
This study explores using iron oxide coatings on glass beads to improve heat transfer efficiency in fluidized bed reactors. Techniques such as BET surface area analysis, SEM imaging, and X-ray diffraction were used to characterize the coated beads. Results showed the successful creation of a crystalline iron layer on the beads’ surface and increased thermal conductivity, especially at elevated temperatures. The study also quantified the impact of air surface velocity and heating power on the heat transfer coefficient, revealing substantial improvements, especially at higher velocities. It was found that the heat transfer coefficient for 600 µm glass beads increases significantly from 336.4 W/m2·K to 390.3 W/m2·K when the velocity is 0.27 m/s and the heating flux is 125 W. This demonstrates the effectiveness of the iron oxide coating in improving heat transfer. The results of this study emphasize the efficacy of iron oxide coatings in augmenting heat transmission characteristics, particularly in fluidized bed reactor.
Full article

Figure 1
Open AccessArticle
Catalysts Based on Iron Oxides for Wastewater Purification from Phenolic Compounds: Synthesis, Physicochemical Analysis, Determination of Catalytic Activity
by
, , , , and
ChemEngineering 2024, 8(1), 8; https://doi.org/10.3390/chemengineering8010008 - 01 Jan 2024
Abstract
►▼
Show Figures
In this work, the synthesis of magnetite nanoparticles and catalysts based on it stabilized with silicon and aluminum oxides was carried out. It is revealed that the stabilization of the magnetite surface by using aluminum and silicon oxides leads to a decrease in
[...] Read more.
In this work, the synthesis of magnetite nanoparticles and catalysts based on it stabilized with silicon and aluminum oxides was carried out. It is revealed that the stabilization of the magnetite surface by using aluminum and silicon oxides leads to a decrease in the size of magnetite nanocrystals in nanocomposites (particle diameter less than ~10 nm). The catalytic activity of the obtained catalysts was evaluated during the oxidation reaction of phenol, pyrocatechin and cresol with oxygen. It is well known that phenolic compounds are among the most dangerous water pollutants. The effect of phenol concentration and the effect of temperature (303–333 K) on the rate of oxidation of phenol to Fe3O4/SiO2 has been studied. It has been determined that the dependence of the oxidation rate of phenol on the initial concentration of phenol in solution is described by a first-order equation. At temperatures of 303–313 K, incomplete absorption of the calculated amount of oxygen is observed, and the analysis data indicate the non-selective oxidation of phenol. Intermediate products, such as catechin, hydroquinone, formic acid, oxidation products, were found. The results of UV and IR spectroscopy showed that catalysts based on magnetite Fe3O4 are effective in the oxidation of phenol with oxygen. In the UV spectrum of the product in the wavelength range 190–1100 nm, there is an absorption band at a wavelength of 240–245 nm and a weak band at 430 nm, which is characteristic of benzoquinone. In the IR spectrum of the product, absorption bands were detected in the region of 1644 cm−1, which is characteristic of the oscillations of the C=O bonds of the carbonyl group of benzoquinone. The peaks also found at 1353 cm−1 and 1229 cm−1 may be due to vibrations of the C-H and C-C bonds of the quinone ring. It was found that among the synthesized catalysts, the Fe3O4/SiO2 catalyst demonstrated the greatest activity in the reaction of liquid-phase oxidation of phenol.
Full article
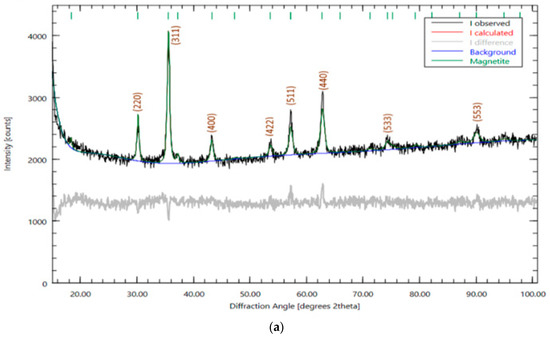
Figure 1
Open AccessArticle
Effect of Inserting Baffles on the Solid Particle Segregation Behavior in Fluidized Bed Reactor: A Computational Study
ChemEngineering 2024, 8(1), 7; https://doi.org/10.3390/chemengineering8010007 - 01 Jan 2024
Abstract
In multi-solid, particle-size fluidized bed reactor systems, segregation is commonly observed. When segregation occurred, small solid particles were entrained to the top of the bed and escaped from the reactor. During the combustion process, the small solid particles that escaped from the boiler
[...] Read more.
In multi-solid, particle-size fluidized bed reactor systems, segregation is commonly observed. When segregation occurred, small solid particles were entrained to the top of the bed and escaped from the reactor. During the combustion process, the small solid particles that escaped from the boiler were burned and subjected to damage around the cyclone separator. This study then employed a computational fluid dynamics approach to investigate solid particle behavior in the reactor using three different sizes of solid particles. The effects of baffle insertion, baffle angle, stage number, and its arrangement were examined. The percentage of segregation was calculated to compare behavior among different reactor systems. The insertion of 45-degree baffles resulted in reduced segregation behavior compared to cases without baffles and with 90-degree baffles, attributed to solid hindering and collision phenomena. Additionally, a double-stage baffle with any arrangement could reduce segregation behavior. The best arrangement was “above-arrangement” due to particles hindering, swirling, and accumulating between the baffle stages. Therefore, to diminish segregation behavior and enhance combustion chemical reactions, the insertion of baffles in the reactor zone is recommended.
Full article
(This article belongs to the Topic Advances in Chemistry and Chemical Engineering)
►▼
Show Figures

Figure 1
Open AccessArticle
Force Field for Calculation of the Vapor-Liquid Phase Equilibrium of trans-Decalin
ChemEngineering 2024, 8(1), 6; https://doi.org/10.3390/chemengineering8010006 - 26 Dec 2023
Abstract
►▼
Show Figures
Based on the TraPPE force field, previously unknown values of the parameters of the intermolecular interaction potential of trans-decalin were determined. Parametrization was carried out using experimental data on saturated vapor pressure and density at atmospheric pressure. The found parameters make it
[...] Read more.
Based on the TraPPE force field, previously unknown values of the parameters of the intermolecular interaction potential of trans-decalin were determined. Parametrization was carried out using experimental data on saturated vapor pressure and density at atmospheric pressure. The found parameters make it possible to adequately describe the boiling and condensation lines of trans-decalin and also predict the critical values of pressure, density, and temperature with satisfactory accuracy. Calculations of vapor-liquid phase equilibrium conditions for a binary CO2—trans-decalin mixture in supercritical conditions for CO2 were carried out. When quantitatively comparing the calculated values with experimental data, an underestimation of pressure at a temperature of 345.4 K by 30% is observed, which decreases to 5% for temperatures up to 525 K.
Full article
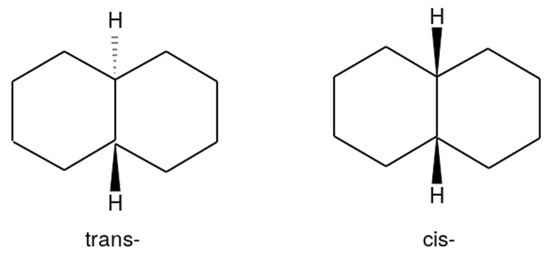
Figure 1
Open AccessArticle
Numerical Simulation of a Valorisation-Oriented Hybrid Process for the Bio-Oil-Related Separation of Acetol and Acetic Acid
ChemEngineering 2024, 8(1), 5; https://doi.org/10.3390/chemengineering8010005 - 22 Dec 2023
Abstract
Biomass as a whole offers a more diverse potential for valorisation than any other renewable energy source. As one of the stages in the separation of bio-oil involves a liquid mixture of acetol and acetic acid, and as both components are particularly well
[...] Read more.
Biomass as a whole offers a more diverse potential for valorisation than any other renewable energy source. As one of the stages in the separation of bio-oil involves a liquid mixture of acetol and acetic acid, and as both components are particularly well suited for valorisation, a hybrid method was developed for their separation with a high purity level through an approach combining liquid–liquid extraction and distillation. In order to design and simulate the flowsheet, the ChemCAD 7.0 simulation software was used. Sensitivity analyses were carried out to investigate the influence of the different parameters in the distillation columns, such as the reflux ratio, the feed stage location, and the vapour/bottom molar flow ratio. The effect of different extractants and of their excess on the separation process, as well as the possibility of regenerating the extractant, was also studied. Tri-n-octylamine was accordingly selected as a separating agent that was fully recycled. The end result for separating an initial 48/52 wt% acetol/acetic acid liquid mixture was acetol with a purity of 99.4 wt% and acetic acid with a purity of 100 wt%.
Full article
(This article belongs to the Special Issue Feature Papers in Chemical Engineering)
►▼
Show Figures
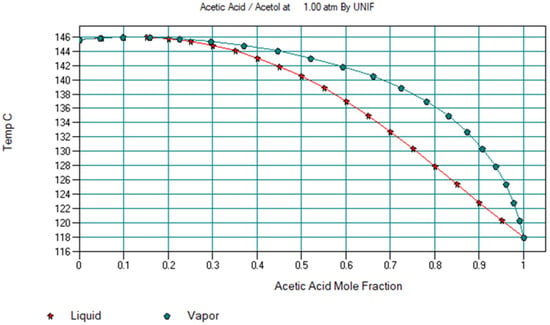
Figure 1
Open AccessArticle
Antisolvent Crystallization of Papain
by
and
ChemEngineering 2024, 8(1), 4; https://doi.org/10.3390/chemengineering8010004 - 20 Dec 2023
Abstract
►▼
Show Figures
Protein crystallization plays a crucial role in the food and pharmaceutical industries, enhancing product quality and efficiency by improving purity and controlled particle characteristics. This study focused on the crystallization of the versatile protein papain, extracted from papaya. Antisolvent crystallization was performed. This
[...] Read more.
Protein crystallization plays a crucial role in the food and pharmaceutical industries, enhancing product quality and efficiency by improving purity and controlled particle characteristics. This study focused on the crystallization of the versatile protein papain, extracted from papaya. Antisolvent crystallization was performed. This method is cost-effective and is a simple and energy-efficient approach. Beyond protein crystal production, the antisolvent crystallization process serves as a method for encapsulating active pharmaceutical ingredients (APIs). The study investigated organic solvents like ethanol, acetone, and acetonitrile as potential antisolvents. Additionally, the impact of variables such as the solvent-to-antisolvent (S:AS) volume ratio and papain concentration on particle size, particle size distribution, zeta potential, crystallization yield, and residual activity of papain crystals were examined. Ethanol emerged as the optimal antisolvent, reducing the solubility of papain and preserving papain’s crystalline structure with minimal activity loss. Optimal conditions were identified at a 1:4 S:AS volume ratio and a papain concentration of 30 mg/mL, resulting in nanosized spherical crystals with a high yield and preserved activity. This research underscored the crucial role of thoughtful parameter selection in antisolvent crystallization to achieve specific particle characteristics while maintaining the functionality of the crystallized substance.
Full article
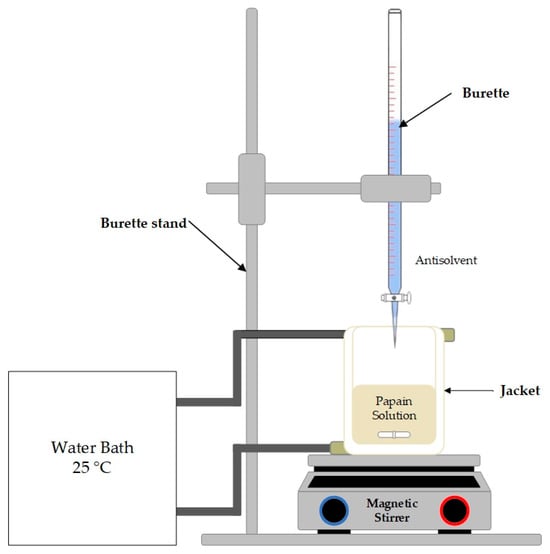
Figure 1
Open AccessCommunication
Ultrafiltration to Increase the Consistency of Fruit Pulps: The Role of Permeate Flux
ChemEngineering 2024, 8(1), 3; https://doi.org/10.3390/chemengineering8010003 - 20 Dec 2023
Abstract
Ultrafiltration is a well-known operation, widely used in food processing, especially to concentrate selectively liquid compounds. However, so far, it has been mainly used to change concentration and/or clarify liquids with low viscosity. Ultrafiltration has seldomly been applied to viscous fluids. In this
[...] Read more.
Ultrafiltration is a well-known operation, widely used in food processing, especially to concentrate selectively liquid compounds. However, so far, it has been mainly used to change concentration and/or clarify liquids with low viscosity. Ultrafiltration has seldomly been applied to viscous fluids. In this study, it was used to increase the consistency of fruit pulps, without changing their taste and organoleptic properties. This paper reports the findings achieved in experimental runs carried out on a pilot plant, equipped with four ultrafiltration tubular membranes (total surface area = 0.8 m2). Raw fruit pulps, namely, apple, apricot, and pear, were used to study the influence of the operative parameters on the permeate flux and organoleptic properties of the final products (retentate and permeate). The flow rate was in the range of 3.0–5.1 m3/h, at 50 °C. The influence of temperature on the permeate flux was checked, with one run with apple pulp at 20 °C. As expected, the findings show that high flow rate and temperature improve the permeate flux. Membranes show different performance in permeate flux for the tested pulps. This is probably due to their different chemical and physical composition, which could be responsible for different fouling of the membrane and, as a consequence, a different resistance to the permeate flow. The final products have the same taste as the raw ones, and each of them can be used as it is or as an ingredient. These results have a technological relevance, and, besides, the study shows a methodology for future applications of ultrafiltration.
Full article
(This article belongs to the Special Issue State-of-the-Art Membrane Technologies in Chemical Engineering)
►▼
Show Figures
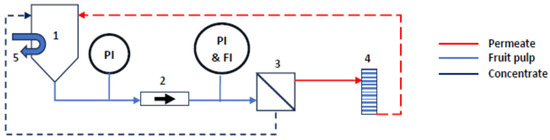
Figure 1
Open AccessArticle
Significant Progress of Initiated Chemical Vapor Deposition in Manufacturing Soft Non-spherical Nanoparticles: Upgrading to the Condensed Droplet Polymerization Approach and Key Technological Aspects
by
ChemEngineering 2024, 8(1), 2; https://doi.org/10.3390/chemengineering8010002 - 19 Dec 2023
Abstract
Initiated chemical vapor deposition is a unique solvent-free and completely dry vapor-phase deposition technique used to synthesize organic polymer films. In this process, an activated initiator, monomer, and carrier gas are introduced into the reaction chamber simultaneously. This technique has been widely adopted.
[...] Read more.
Initiated chemical vapor deposition is a unique solvent-free and completely dry vapor-phase deposition technique used to synthesize organic polymer films. In this process, an activated initiator, monomer, and carrier gas are introduced into the reaction chamber simultaneously. This technique has been widely adopted. However, if the monomer and initiator are introduced into the chamber in stages—allowing gas-phase monomer deposition and condensation first, followed by initiator introduction and controlling the monomer partial pressure to be higher than the saturated vapor pressure—non-spherical polymer nanoparticles with dome-like shapes can be obtained. This advanced iCVD technique is referred to as the “Condensed Droplet Polymerization Approach”. This high monomer partial pressure gas-phase deposition is not suitable for forming uniformly composed iCVD films; but interestingly, it can rapidly obtain polymer nanodomes (PNDs). Using CDP technology, Franklin polymerized multifunctional nanodomes in less than 45 s, demonstrating a wide range of continuous particle size variations, from sub-20 nanometers to over 1 micron. This rapid synthesis included a variety of functional polymer nanodomes in just a matter of seconds to minutes. This review discusses the crucial process conditions of the Condensed Droplet Polymerization (CDP) Approach for synthesizing PNDs. The main focus of the discussion was on the two-step method for synthesizing PNDs, where the nucleation mechanism of PNDs, factors influencing their size, and the effect of pressure on the distinct condensation of monomer vapor into polymer nanodomes and polymer films were extensively explored.
Full article
(This article belongs to the Topic Advances in Chemistry and Chemical Engineering)
►▼
Show Figures
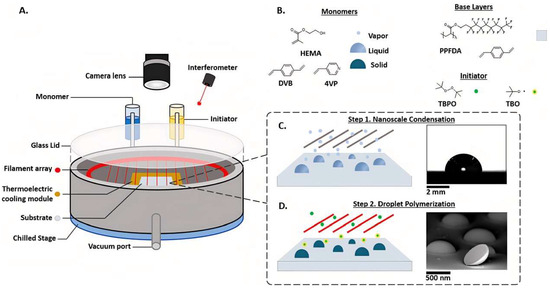
Figure 1
Open AccessArticle
Improved Fault Detection in Chemical Engineering Processes via Non-Parametric Kolmogorov–Smirnov-Based Monitoring Strategy
ChemEngineering 2024, 8(1), 1; https://doi.org/10.3390/chemengineering8010001 - 19 Dec 2023
Abstract
►▼
Show Figures
Fault detection is crucial in maintaining reliability, safety, and consistent product quality in chemical engineering processes. Accurate fault detection allows for identifying anomalies, signaling deviations from the system’s nominal behavior, ensuring the system operates within desired performance parameters, and minimizing potential losses. This
[...] Read more.
Fault detection is crucial in maintaining reliability, safety, and consistent product quality in chemical engineering processes. Accurate fault detection allows for identifying anomalies, signaling deviations from the system’s nominal behavior, ensuring the system operates within desired performance parameters, and minimizing potential losses. This paper presents a novel semi-supervised data-based monitoring technique for fault detection in multivariate processes. To this end, the proposed approach merges the capabilities of Principal Component Analysis (PCA) for dimensionality reduction and feature extraction with the Kolmogorov–Smirnov (KS)-based scheme for fault detection. The KS indicator is computed between the two distributions in a moving window of fixed length, allowing it to capture sensitive details that enhance the detection of faults. Moreover, no labeling is required when using this fault detection approach, making it flexible in practice. The performance of the proposed PCA–KS strategy is assessed for different sensor faults on benchmark processes, specifically the Plug Flow Reactor (PFR) process and the benchmark Tennessee Eastman (TE) process. Different sensor faults, including bias, intermittent, and aging faults, are considered in this study to evaluate the proposed fault detection scheme. The results demonstrate that the proposed approach surpasses traditional PCA-based methods. Specifically, when applied to PFR data, it achieves a high average detection rate of 98.31% and a low false alarm rate of 0.25%. Similarly, when applied to the TE process, it provides a good average detection rate of 97.27% and a false alarm rate of 6.32%. These results underscore the efficacy of the proposed PCA–KS approach in enhancing the fault detection of high-dimensional processes.
Full article
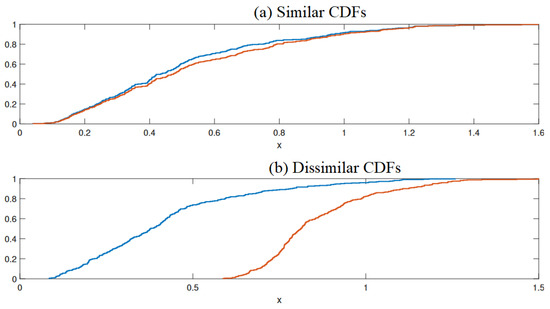
Figure 1
Open AccessArticle
Microwave-Assisted Synthesis of Titanosilicates Using a Precursor Produced from Titanium Ore Concentrate
by
, , , , , , , , , , , , , and
ChemEngineering 2023, 7(6), 118; https://doi.org/10.3390/chemengineering7060118 - 15 Dec 2023
Abstract
►▼
Show Figures
Titanosilicates comprise a broad class of materials with promising technological applications. The typical obstacle that restricts their industrial applicability is the high manufacturing cost due to the use of specific organotitanium precursors. We herein report a new approach to the synthesis of titanosilicates
[...] Read more.
Titanosilicates comprise a broad class of materials with promising technological applications. The typical obstacle that restricts their industrial applicability is the high manufacturing cost due to the use of specific organotitanium precursors. We herein report a new approach to the synthesis of titanosilicates using an inexpensive inorganic precursor, ammonium titanyl sulfate (ATS or STA), (NH4)2TiO(SO4)2∙H2O. The latter is an intermediate in the processing of titanium-bearing concentrates produced from apatite-nepheline ores. In this paper, the new synthetic approach is exemplified by the microwave-assisted synthesis of IONSIVE-911, one of the most effective Cs-ion scavengers. The method can be modified to synthesize various titanosilicate compounds.
Full article
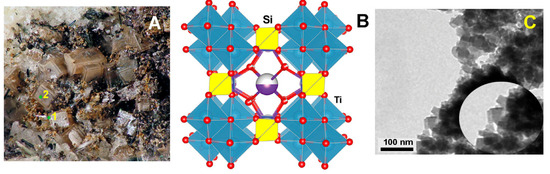
Figure 1
Open AccessArticle
Evaluation of Stabilization and Physical–Chemical Properties of CNT Antifreeze Nanofluid Prepared in 50:50 EG/Water by Modified Strategy
ChemEngineering 2023, 7(6), 117; https://doi.org/10.3390/chemengineering7060117 - 06 Dec 2023
Abstract
►▼
Show Figures
This article proposes a better alternative method to prepare CNT antifreeze nanofluid in EG/water by modifying the conventional method that requires long hours of sonication. Sonicating a sample for long hours is time and energy consuming and may deform the structure of CNT.
[...] Read more.
This article proposes a better alternative method to prepare CNT antifreeze nanofluid in EG/water by modifying the conventional method that requires long hours of sonication. Sonicating a sample for long hours is time and energy consuming and may deform the structure of CNT. In the modified method, the nanofluid preparation was carried out by dispersion of CNT in EG via sonication followed by adding water and again sonication. The study shows that nanofluid could be prepared in less sonication time of 1.5 h compared to the 5 h required in the conventional method. FTIR spectroscopy revealed that interaction of EG with CNT occurs via trans conformation resulting in greater stabilization and better interaction of nanofluid prepared by this method (85 days) as compared to nanofluid prepared by the conventional method (50 days). The nanofluid prepared by this method has better physical–chemical properties compared to nanofluid prepared by the conventional method. The nanofluid prepared by this method showed higher stability and better physical–chemical properties at a lower sonication time. Hence it is a more effective and cost efficient technique for preparing CNT (EG/water) nanofluid.
Full article
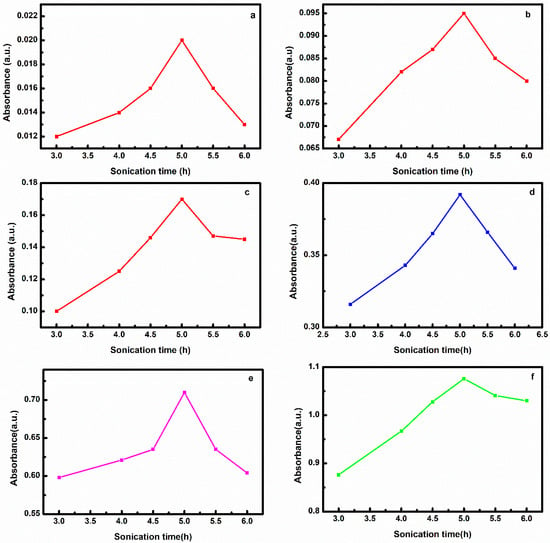
Figure 1
Open AccessReview
Exploring Strategies for Copper Removal from Nickel Anolytes: A Review
by
and
ChemEngineering 2023, 7(6), 116; https://doi.org/10.3390/chemengineering7060116 - 05 Dec 2023
Abstract
►▼
Show Figures
Various methods, such as electrochemical purification, chemical precipitation, solvent extraction, and ion-exchange resins, have been extensively employed for the removal of copper from nickel anolytes. However, these methods exhibit several significant drawbacks when applied in industrial settings. For instance, electrochemical purification fails to
[...] Read more.
Various methods, such as electrochemical purification, chemical precipitation, solvent extraction, and ion-exchange resins, have been extensively employed for the removal of copper from nickel anolytes. However, these methods exhibit several significant drawbacks when applied in industrial settings. For instance, electrochemical purification fails to efficiently manage nickel anolyte solutions with low copper content. Chemical precipitation presents challenges in residue management and incurs high production costs for precipitants. Solvent extraction raises concerns related to toxicity, while the use of ion-exchange resins demands meticulous selection of suitable materials. In this review, we present a comprehensive review of the nickel removal methods used for nickel anolyte purification, electrochemical purification, chemical precipitation, solvent extraction, and ion-exchange resins. We also examine the suitability and benefits of each technique in industrial settings. The ion-exchange method has drawn significant attention due to its strong selectivity and small adsorption quantity. The ion-exchange separation process does not generate any slag, and the ion-exchange resin can be recycled and reused; this method has great potential in a wide range of applications.
Full article
Figure 1
Open AccessArticle
Experimental and Kinetic Study of the Catalytic Behavior of Sulfate-Treated Nanostructured Bifunctional Zirconium Oxide Catalysts in n-Heptane Hydroisomerization Reactions
ChemEngineering 2023, 7(6), 115; https://doi.org/10.3390/chemengineering7060115 - 01 Dec 2023
Abstract
►▼
Show Figures
In this study, a mono-functional ZrO2 nanomaterial was treated with sulfur and loaded with two different percentages of platinum metals (i.e., 0.5 and 1 wt%) to generate an acidic bi-functional Pt/SZrO2 nanocatalyst for the purpose of increasing the catalytic activity and
[...] Read more.
In this study, a mono-functional ZrO2 nanomaterial was treated with sulfur and loaded with two different percentages of platinum metals (i.e., 0.5 and 1 wt%) to generate an acidic bi-functional Pt/SZrO2 nanocatalyst for the purpose of increasing the catalytic activity and selectivity together. This work aims to determine the least amount of the costly platinum metal that can be added to the catalyst to achieve the appropriate balance between the acidic and metallic sites. Both rapid deactivation of the super-acid nanaocatalyst and fast cleavage of the zero-octane n-heptane chain can consequently be prevented throughout the reaction. This can be achieved by accelerating the hydroisomerization reactions at a pressure of 5 bar to reach the highest selectivity towards producing the desired multi-branched compound in fuel. Several characterization techniques, including XRD, SEM, EDX, BET, and FTIR, have been used to evaluate the physical properties of the catalysts. The best reaction product was obtained at 230 °C compared to the other tested temperatures. The conversion, selectivity, and yield of reaction products over the surfaces of the prepared catalysts followed this order: 0.5 wt% Pt/SZrO2 > 1 wt% Pt/SZrO2 > 0.5 wt% Pt/ZrO2 > 1 wt% Pt/ZrO2 > SZrO2 > ZrO2. The highest conversion, selectivity, and yield values were obtained on the surface of the 0.5 wt% Pt/SZrO2 catalyst, which are 69.64, 81.4 and 56.68 wt%, respectively, while the lowest values were obtained on the surface of the parent ZrO2 catalyst, which are 43.9, 61.1 and 26.82, respectively. The kinetic model and apparent activation energies were also implemented for each of the hydroisomerization, hydrogenation/dehydrogenation, and hydrocracking reactions, which track the following order: hydroisomerization < hydrogenation/dehydrogenation < hydrocracking. The lowest apparent activation energy value of 123.39 kJ/mol was found on the surface of the most active and selective 0.5% Pt/SZrO2 nanocatalyst.
Full article
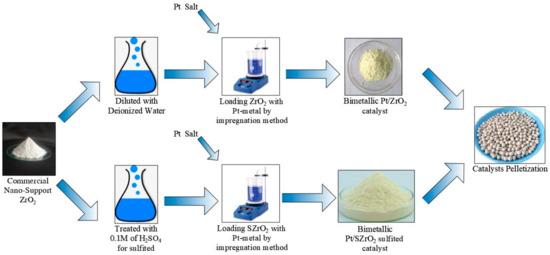
Figure 1
Open AccessArticle
Methyl Orange Adsorption on Biochar Obtained from Prosopis juliflora Waste: Thermodynamic and Kinetic Study
ChemEngineering 2023, 7(6), 114; https://doi.org/10.3390/chemengineering7060114 - 01 Dec 2023
Abstract
►▼
Show Figures
In the information contained herein, we fabricated biochar by means of a pyrolysis process; it used Prosopis juliflora waste (PJW) as a biomass source. The physical and chemical material characterization was carried out through FTIR, thermogravimetric, BET-N2 isotherm, and SEM-EDX assays. We
[...] Read more.
In the information contained herein, we fabricated biochar by means of a pyrolysis process; it used Prosopis juliflora waste (PJW) as a biomass source. The physical and chemical material characterization was carried out through FTIR, thermogravimetric, BET-N2 isotherm, and SEM-EDX assays. We studied the methylene orange (MO) adsorption onto PWJ biochar. The PJW biochar displayed a maximum percentage of MO removal of 64%. The results of the adsorption study indicated that Temkin isotherm was suitable to describe the MO adsorption process on PJW biochar; it suggests that the MO adsorption on PJW biochar could be a multi-layer adsorption process. Results showed that the pseudo-second-order model was accurate in demonstrating the MO adsorption on PJW (k2 = 0.295 g mg−1min−1; qe = 8.31 mg g−1). Furthermore, the results made known that the MO removal by PJW biochar was endothermic (ΔH = 12.7 kJ/mol) and a spontaneous process (ΔG = −0.954 kJ/mol). The reusability test disclosed that after four consecutive adsorption/desorption cycles, the PWJ biochar reduced its MO removal by only 4.3%.
Full article
Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Energies, Processes, Bioengineering, ChemEngineering, Clean Technol.
Chemical and Biochemical Processes for Energy Sources
Topic Editors: Venko N. Beschkov, Konstantin PetrovDeadline: 31 January 2024
Topic in
AppliedChem, ChemEngineering, Energies, Membranes, Processes, Recycling, Separations, Water
Capacitive Deionization Technology for Water Treatment
Topic Editors: Shenxu Bao, Xin ZhangDeadline: 30 September 2024

Conferences
Special Issues
Special Issue in
ChemEngineering
Catalytic Reactions and Development of (Bio)Chemical Processes for Synthesizing Value Added Compounds
Guest Editor: Ionut BanuDeadline: 31 January 2024
Special Issue in
ChemEngineering
Advances in Catalytic Kinetics
Guest Editors: Dmitry Murzin, Philippe HeynderickxDeadline: 20 February 2024
Special Issue in
ChemEngineering
Fueling the Future: Chemical Engineering Approaches in Ceramic Materials for Energy Storage
Guest Editors: Jayanthi Kumar, Min NiuDeadline: 31 March 2024
Special Issue in
ChemEngineering
Emerging Technologies for Remediation of per- and Polyfluoroalkyl Substances (PFAS) from Contaminated Water
Guest Editors: Debarati Mukherjee, Biswajit Bera, Sirshendu DeDeadline: 30 April 2024
Topical Collections
Topical Collection in
ChemEngineering
Green and Environmentally Sustainable Chemical Processes
Collection Editors: Roberto Rosa, Anna Ferrari, Consuelo Mugoni, Grazia Maria Cappucci








.jpg)







