Journal Description
Corrosion and Materials Degradation
Corrosion and Materials Degradation
is an international, peer-reviewed, open access journal on corrosion, environment-assisted degradation, corrosion mitigation, corrosion mechanism and corrosion monitoring, published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, EBSCO, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 20.2 days after submission; acceptance to publication is undertaken in 5.6 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
Latest Articles
Microbial Communities in Model Seawater-Compensated Fuel Ballast Tanks: Biodegradation and Biocorrosion Stimulated by Marine Sediments
Corros. Mater. Degrad. 2024, 5(1), 1-26; https://doi.org/10.3390/cmd5010001 - 03 Jan 2024
Abstract
►
Show Figures
Some naval vessels add seawater to carbon steel fuel ballast tanks to maintain stability during fuel consumption. Marine sediments often contaminate ballast tank fluids and have been implicated in stimulating fuel biodegradation and enhancing biocorrosion. The impact of the marine sediment was evaluated
[...] Read more.
Some naval vessels add seawater to carbon steel fuel ballast tanks to maintain stability during fuel consumption. Marine sediments often contaminate ballast tank fluids and have been implicated in stimulating fuel biodegradation and enhancing biocorrosion. The impact of the marine sediment was evaluated in model ballast tank reactors containing seawater, fuel (petroleum-F76, Fischer–Tropsch F76, or a 1:1 mixture), and carbon steel coupons. Control reactors did not receive fuel. The marine sediment was added to the reactors after 400 days and incubated for another year. Sediment addition produced higher estimated bacterial numbers and enhanced sulfate reduction. Ferrous sulfides were detected on all coupons, but pitting corrosion was only identified on coupons exposed to FT-F76. Aerobic hydrocarbon-degrading bacteria increased, and the level of dissolved iron decreased, consistent with the stimulation of aerobic hydrocarbon degradation by iron. We propose that sediments provide an inoculum of hydrocarbon-degrading microbes that are stimulated by dissolved iron released during steel corrosion. Hydrocarbon degradation provides intermediates for use by sulfate-reducing bacteria and reduces the level of fuel components inhibitory to anaerobic bacteria. The synergistic effect of dissolved iron produced by corrosion, biodegradable fuels, and iron-stimulated hydrocarbon-degrading microbes is a poorly recognized but potentially significant biocorrosion mechanism.
Full article
Open AccessArticle
Data Mining Applied to the Electrochemical Noise Technique in the Time/Frequency Domain for Stress Corrosion Cracking Recognition
Corros. Mater. Degrad. 2023, 4(4), 659-679; https://doi.org/10.3390/cmd4040034 - 06 Dec 2023
Abstract
In this paper, time/frequency domain data processing was proposed to analyse the EN signal recorded during stress corrosion cracking on precipitation-hardening martensitic stainless steel in a chloride environment. Continuous Wavelet Transform, albeit with some limitations, showed a suitable support in the discriminatory capacity
[...] Read more.
In this paper, time/frequency domain data processing was proposed to analyse the EN signal recorded during stress corrosion cracking on precipitation-hardening martensitic stainless steel in a chloride environment. Continuous Wavelet Transform, albeit with some limitations, showed a suitable support in the discriminatory capacity among transient signals related to the different stress corrosion cracking mechanisms. In particular, the aim is to propose the analysis of electrochemical noise signals under stress corrosion cracking conditions in the time–frequency domain by using the Hilbert–Huang approach. The Hilbert–Huang Transform (performed by the Empirical Mode Decomposition approach) was finally proposed to carry out an identification of the corrosion mechanisms in comparison to conventional data processing methods. By using this approach, a detailed simultaneous decomposition of the original electrochemical noise data in the time and frequency domain was carried out. The method gave useful information about transitions among different corrosion mechanisms, allowing us to (i) identify a specific characteristic response for each corrosion damaging phenomenon induced by stress corrosion cracking, (ii) time each corrosion of the damaging phenomenon, and (iii) provide a topological description of the advancing SCC damaging stages. This characteristic evidences that the Hilbert–Huang Transform is a very powerful technique to potentially recognize and distinguish the different corrosion mechanisms occurring during stress corrosion cracking.
Full article
(This article belongs to the Special Issue Exclusive Papers Collection of Editorial Board Members of Corrosion and Materials Degradation 2023)
►▼
Show Figures
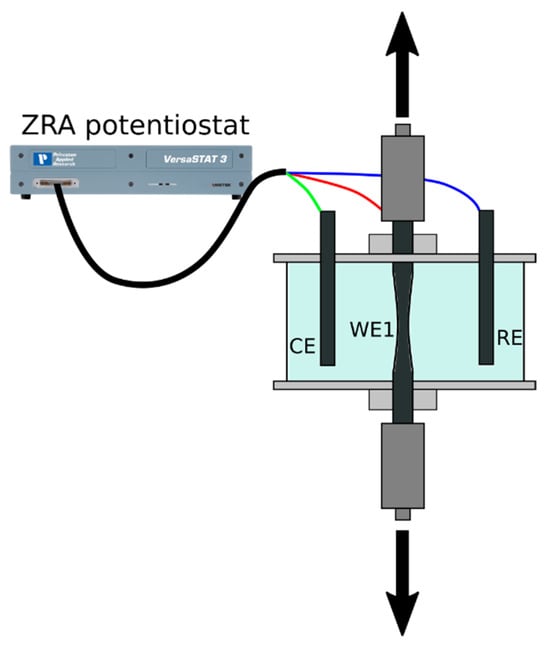
Figure 1
Open AccessReview
Review on Corrosion, Tribocorrosion and Osseointegration of Titanium Alloys as Biomaterials
Corros. Mater. Degrad. 2023, 4(4), 644-658; https://doi.org/10.3390/cmd4040033 - 28 Nov 2023
Abstract
►▼
Show Figures
When introduced into the body, the implant interacts with biological environment and may suffer corrosion. In addition, when this implant is submitted to friction, it may degrade by tribocorrosion due to the simultaneous action of corrosion by the body liquid and mechanical wear.
[...] Read more.
When introduced into the body, the implant interacts with biological environment and may suffer corrosion. In addition, when this implant is submitted to friction, it may degrade by tribocorrosion due to the simultaneous action of corrosion by the body liquid and mechanical wear. Both corrosion and tribocorrosion are connected to the presence of proteins that cover the surface implant. The latter plays an ambiguous role on corrosion since dozens of contradictory papers pointed out their beneficial or detrimental effect. After its introduction into the body, the implant should form a direct interface with bone through structural and functional connection. The osseointegration and the strength of interfacial bond depend on surface properties of the implant, namely, its topographical and physico-chemical properties. In addition, since bone cells are sensitive to the species produced during the implant corrosion, when corrosion occurs, this may lead to impact osseointegration and to cause implant loosening. There is a strong connection between corrosion and osseointegration, both of which are worth discussion. That is the object of the present narrative review where we will discuss: (1) corrosion and tribocorrosion of titanium alloys used as biomaterials paying particular attention to the influence of proteins, (2) the effect of implant roughness and surface energy on osseointegration.
Full article
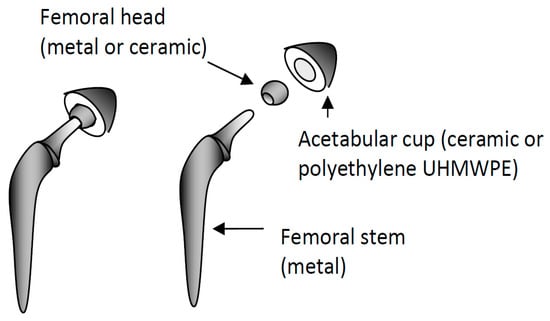
Figure 1
Open AccessArticle
Improving the Mechanical and Electrochemical Performance of Additively Manufactured 8620 Low Alloy Steel via Boriding
Corros. Mater. Degrad. 2023, 4(4), 623-643; https://doi.org/10.3390/cmd4040032 - 06 Nov 2023
Cited by 1
Abstract
►▼
Show Figures
In this study, mechanical and electrochemical performance of borided additively manufactured (AM) and wrought 8620 low alloy steel were investigated and compared to their bare counterparts. The microstructure of borided 8620 exhibited the presence of FeB and Fe2B phases with a
[...] Read more.
In this study, mechanical and electrochemical performance of borided additively manufactured (AM) and wrought 8620 low alloy steel were investigated and compared to their bare counterparts. The microstructure of borided 8620 exhibited the presence of FeB and Fe2B phases with a saw tooth morphology. Both AM and wrought samples with boride layers showed a similar performance in hardness, wear, potentiodynamic polarization (PD), electrochemical impedance spectroscopy (EIS), and linear polarization resistance (LPR) experiments. However, borided steels exhibited about an 8-fold increase in Vickers hardness and about a 6-fold enhancement in wear resistance compared to bare ones. Electrochemical experiments of borided specimens (both AM and wrought) in 0.1 M Na2S2O3 + 1 M NH4Cl solution revealed a 3–6-fold lower corrosion current density, about a 6-fold higher charge transfer resistance, and about a 6-fold lower double-layer capacitance, demonstrating an improved corrosion resistance compared to their bare counterparts. Post-corrosion surface analysis revealed the presence of thick sulfide and oxide layers on the bare steels, whereas dispersed corrosion particles were observed on the borided samples. The enhanced wear and electrochemical performance of the borided steels were attributed to the hard FeB/Fe2B layers and the reduced amount of adsorbed sulfur on their surface.
Full article
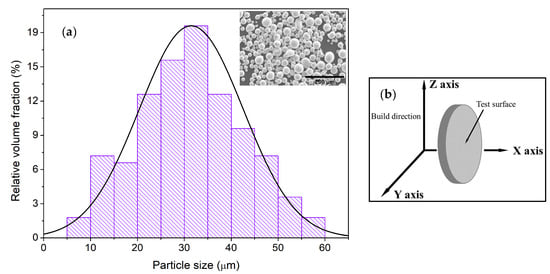
Figure 1
Open AccessReview
A Review on Tribocorrosion Behavior of Aluminum Alloys: From Fundamental Mechanisms to Alloy Design Strategies
Corros. Mater. Degrad. 2023, 4(4), 594-622; https://doi.org/10.3390/cmd4040031 - 18 Oct 2023
Cited by 1
Abstract
Tribocorrosion, a research field that has been evolving for decades, has gained renewed attention in recent years, driven by increased demand for wear- and corrosion-resistant materials from biomedical implants, nuclear power generation, advanced manufacturing, batteries, marine and offshore industries, etc. In the United
[...] Read more.
Tribocorrosion, a research field that has been evolving for decades, has gained renewed attention in recent years, driven by increased demand for wear- and corrosion-resistant materials from biomedical implants, nuclear power generation, advanced manufacturing, batteries, marine and offshore industries, etc. In the United States, wear and corrosion are estimated to cost nearly USD 300 billion per year. Among various important structural materials, passive metals such as aluminum alloys are most vulnerable to tribocorrosion due to the wear-accelerated corrosion as a result of passive film removal. Thus, designing aluminum alloys with better tribocorrosion performance is of both scientific and practical importance. This article reviews five decades of research on the tribocorrosion of aluminum alloys, from experimental to computational studies. Special focus is placed on two aspects: (1) The effects of alloying and grain size on the fundamental wear, corrosion, and tribocorrosion mechanisms; and (2) Alloy design strategies to improve the tribocorrosion resistance of aluminum alloys. Finally, the paper sheds light on the current challenges faced and outlines a few future research directions in the field of tribocorrosion of aluminum alloys.
Full article
(This article belongs to the Special Issue Mechanism and Predictive/Deterministic Aspects of Corrosion)
►▼
Show Figures
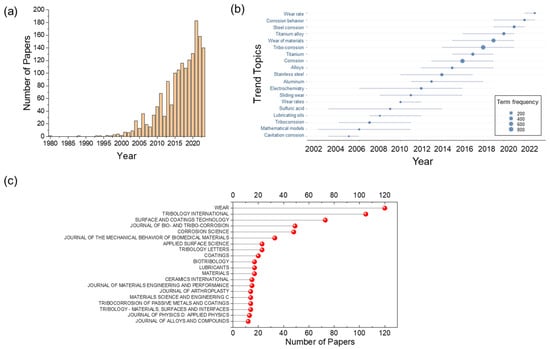
Figure 1
Open AccessArticle
Evaluating the Impact of Redox Potential on the Corrosion of Q125, 316L, and C276 Steel in Low-Temperature Geothermal Systems
Corros. Mater. Degrad. 2023, 4(4), 573-593; https://doi.org/10.3390/cmd4040030 - 08 Oct 2023
Abstract
►▼
Show Figures
Time series experiments were used to explore the fluid redox impact on the corrosion of Q125, 316L, and C276 steels in low-ionic-strength and neutral water at temperature and pressure conditions associated with low-temperature geothermal systems. After exposing polished samples of each steel grade
[...] Read more.
Time series experiments were used to explore the fluid redox impact on the corrosion of Q125, 316L, and C276 steels in low-ionic-strength and neutral water at temperature and pressure conditions associated with low-temperature geothermal systems. After exposing polished samples of each steel grade to an oxidizing (H2O2) and a reducing (Zn-doped) fluid for intervals of 24 h, 1 week, and 6 weeks, the atomic force microscopy results revealed general corrosion for Q125, while 316L and C276 exhibited pitting, crevice expansion, and edge attack corrosion. Secondary depositional features are frequently found as topographic highs, adjacent to pitting corrosion. These features may be identified as there is a very strong spatial correlation between the height retrace and phase retrace surface maps. All steels became progressively rougher over time after exposure to both fluids, while the corrosion rates were more complex. Samples exposed to the reducing fluid experienced an increase in the corrosion rate over time, while C276 and 316L experienced a decrease in the corrosion rate. Finally, a novel data validation technique was developed to address the intrinsic scalability of corrosion. The results indicate that the AFM scan area does not affect the measured surface roughness over nearly three orders of magnitude.
Full article
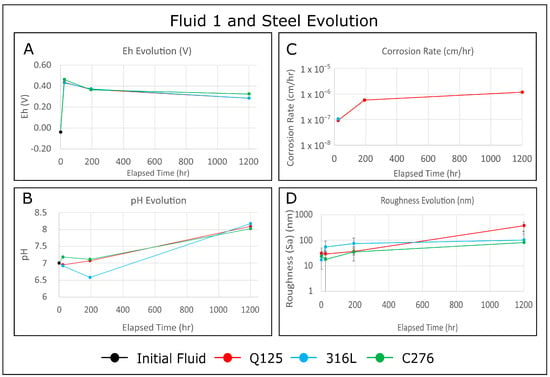
Figure 1
Open AccessFeature PaperReview
State-of-the-Art Review of Aliphatic Polyesters and Polyolefins Biodeterioration by Microorganisms: From Mechanism to Characterization
Corros. Mater. Degrad. 2023, 4(4), 542-572; https://doi.org/10.3390/cmd4040029 - 04 Oct 2023
Abstract
►▼
Show Figures
As a result of the exponential growth in the production of plastics and their extended degradation period, strong environmental concerns in association with the disposal of plastic waste have emerged. Pursuing sustainable solutions for managing plastic waste has led to significant interest in
[...] Read more.
As a result of the exponential growth in the production of plastics and their extended degradation period, strong environmental concerns in association with the disposal of plastic waste have emerged. Pursuing sustainable solutions for managing plastic waste has led to significant interest in plastic biodegradation research, with a specific focus on biodeterioration facilitated by microorganisms. The biodeterioration of plastic by microorganisms is a complex phenomenon that can be influenced by a variety of environmental factors such as humidity, temperature, and pH, as well as polymer properties such as molecular structure, molecular weight, and crystallinity. Toward a better understanding of this phenomenon for resolving the issue of plastic waste, this review article focuses on the biodeterioration of synthetic polymers, in particular aliphatic polyesters and polyolefins, through the enzymatic activities of microorganisms. First, the mechanism of polymer biodegradation via enzymatic activity is discussed, followed by the physical properties of polymers and environmental conditions that influence their biodegradability rates. Then, an overview of experimental approaches and standardized protocols used to assess the biodegradability of polymers by these degrading agents is provided. Finally, current developments in employing biodeterioration for the degradation of aliphatic polyesters and polyolefins are reviewed. The review concludes with a discussion on the complexity of biodegradation by microorganisms, the necessity of proper engineering of polymer properties during production to enhance their biodegradability, and the need for further research to discover sustainable and environmentally acceptable alternatives.
Full article
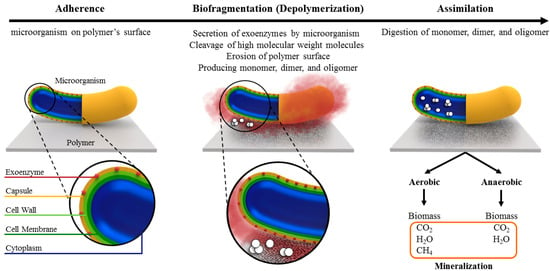
Figure 1
Open AccessArticle
Oxidation Behavior at 1000 °C of Low-Mn High-Cr Cantor’s HEA-Based Alloys Strengthened or Not by MC Carbides
by
, , , , , , and
Corros. Mater. Degrad. 2023, 4(4), 528-541; https://doi.org/10.3390/cmd4040028 - 25 Sep 2023
Abstract
►▼
Show Figures
A conventionally cast CoNiFeMn0.5Cr1.5 alloy and two versions with 0.25 C & 3.7 Ta or 0.25 C & 3.7 Hf were tested in oxidation at 1000 °C for 50 h with thermogravimetric recording of the oxidation kinetic. In all cases,
[...] Read more.
A conventionally cast CoNiFeMn0.5Cr1.5 alloy and two versions with 0.25 C & 3.7 Ta or 0.25 C & 3.7 Hf were tested in oxidation at 1000 °C for 50 h with thermogravimetric recording of the oxidation kinetic. In all cases, the obtained mass gain curve is parabolic. The parabolic constants are much lower than the Kp previously determined for the original alloys with an equimolar base (CoNiFeMnCr). However, the post-mortem exploitation of the oxidized samples revealed analogous oxidation features on the surface and the subsurface, also with external oxide strata on the surface with different Mn and Cr contents, and rather great Mn depletion, in addition to a moderate Cr depletion, in the subsurface. Globally, the oxidation behavior is significantly better than was earlier observed for the equimolar version of these alloys.
Full article

Figure 1
Open AccessArticle
Self-Healing Coatings Consisting of an Outer Electrodeposited Epoxy Resin Layer and an Inner Porous Anodic Oxide Layer with Healing Agents for the Corrosion Protection of Al Alloys
Corros. Mater. Degrad. 2023, 4(3), 516-527; https://doi.org/10.3390/cmd4030027 - 18 Sep 2023
Abstract
►▼
Show Figures
Recently, new surface treatments for the corrosion protection of Al alloys by forming self-healing layers have attracted the attention of many researchers. The authors of this paper have previously developed self-healing polyurethane coatings with micro-capsules containing healing agents and porous anodic oxide films
[...] Read more.
Recently, new surface treatments for the corrosion protection of Al alloys by forming self-healing layers have attracted the attention of many researchers. The authors of this paper have previously developed self-healing polyurethane coatings with micro-capsules containing healing agents and porous anodic oxide films filled with healing agents. In this study, self-healing coatings consisting of an outer electrodeposited epoxy resin layer and an inner porous anodic oxide layer with healing agents were developed for the corrosion protection of Al alloys. The corrosion protection abilities of the self-healing coating were shown in Cu2+/Cl− solutions after damaging with indenters and were affected by freezing treatments and the tip angles of the indenter.
Full article
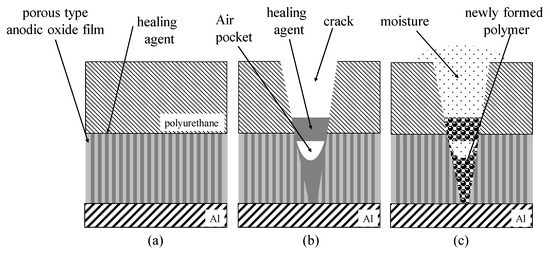
Figure 1
Open AccessArticle
The Effect of Microstructure on Local Corrosion Product Formation during Initial SO2-Induced Atmospheric Corrosion of ZnAlMg Coating Studied by FTIR-ATR FPA Chemical Imaging
Corros. Mater. Degrad. 2023, 4(3), 503-515; https://doi.org/10.3390/cmd4030026 - 08 Sep 2023
Abstract
The initial atmospheric corrosion of ZM (ZnAlMg)-coated steel in humid air (85% RH) and humid argon (85% RH) containing 320 ppb SO2 was studied using in situ infrared reflection absorption spectroscopy (IRRAS), FTIR-ATR focal plane array (FPA) imaging and SEM-EDS. The corrosion
[...] Read more.
The initial atmospheric corrosion of ZM (ZnAlMg)-coated steel in humid air (85% RH) and humid argon (85% RH) containing 320 ppb SO2 was studied using in situ infrared reflection absorption spectroscopy (IRRAS), FTIR-ATR focal plane array (FPA) imaging and SEM-EDS. The corrosion products formed in humid air containing SO2 are mainly composed of magnesium sulphites and sulphates, with sulphite-containing corrosion products formed initially while the contribution from sulphates increased with exposure time. The results from FTIR-FPA imaging and SEM-EDS showed that the magnesium sulphite and sulphate are formed mainly on eutectic phases with a higher quantity of corrosion products formed on the binary eutectic (Zn-MgZn2) phases. This is due to presence of microgalvanic elements with the zinc-rich phases as the main sites for the cathodic oxygen reduction while the anodic reactions take place on the eutectic areas. Sulphate content is the highest on the binary eutectic phases, due to the microgalvanic effects and the production of oxidants by the cathodic reaction, which increases the oxidation of sulphite to sulphate.
Full article
(This article belongs to the Special Issue Atmospheric Corrosion of Materials)
►▼
Show Figures

Figure 1
Open AccessFeature PaperArticle
Plasma Electrolytic Oxidation on Magnesium AZ31 with Sepiolite as Inhibitor Carrier for Improved Corrosion Protection
by
, , , , and
Corros. Mater. Degrad. 2023, 4(3), 488-502; https://doi.org/10.3390/cmd4030025 - 30 Aug 2023
Abstract
►▼
Show Figures
Plasma electrolytic oxidation (PEO) in an alkaline silicate electrolyte containing nanosized sepiolite fibers was carried out on magnesium alloy AZ31. The mineral fibers were loaded with different corrosion inhibitors and incorporated in situ during the PEO treatment. The composition and microstructure of the
[...] Read more.
Plasma electrolytic oxidation (PEO) in an alkaline silicate electrolyte containing nanosized sepiolite fibers was carried out on magnesium alloy AZ31. The mineral fibers were loaded with different corrosion inhibitors and incorporated in situ during the PEO treatment. The composition and microstructure of the PEO coatings were investigated by SEM. It was shown that the fibers are located on the surface as well as inside the “weak spots” of the coating, i.e., pores and discharge channels. The fixation of the particles is caused by sintering due to the heat developed during the PEO treatment. Investigations using electrochemical impedance spectroscopy and linear sweep voltammetry in 0.01 M NaCl solution confirmed an improvement of the corrosion protection. The use of the inhibitors shifts the critical pitting potential in the anodic direction. Regarding efficiency, cerium-loaded sepiolite showed the best behavior by shifting the pitting potential by +0.9 V.
Full article
Figure 1
Open AccessFeature PaperReview
Improved and Innovative Accident-Tolerant Nuclear Fuel Materials Considered for Retrofitting Light Water Reactors—A Review
Corros. Mater. Degrad. 2023, 4(3), 466-487; https://doi.org/10.3390/cmd4030024 - 24 Aug 2023
Cited by 1
Abstract
Since 2011, there has been an international effort to evaluate the behavior of newer fuel rod materials for the retrofitting of existing light water reactors (LWR). These materials include concepts for the cladding of the fuel and for the fuel itself. The materials
[...] Read more.
Since 2011, there has been an international effort to evaluate the behavior of newer fuel rod materials for the retrofitting of existing light water reactors (LWR). These materials include concepts for the cladding of the fuel and for the fuel itself. The materials can be broadly categorized into evolutionary or improved existing materials and revolutionary or innovative materials. The purpose of the newer materials or accident-tolerant fuels (ATF) is to make the LWRs more resistant to loss-of-coolant accidents and thus increase their operation safety. The benefits and detriments of the three main concepts for the cladding are discussed. These include (i) coatings for existing zirconium alloys; (ii) monolithic iron–chromium–aluminum alloys; and (iii) composites based on silicon carbide. The use of ATF materials may help extend the life of currently operating LWRs, while also being a link to material development for future commercial reactors.
Full article
(This article belongs to the Special Issue Mechanism and Predictive/Deterministic Aspects of Corrosion)
►▼
Show Figures
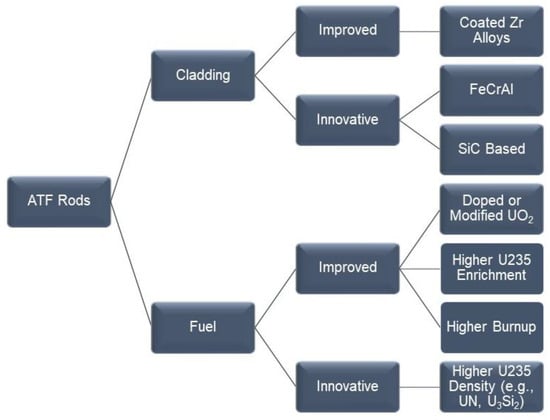
Figure 1
Open AccessArticle
Corrosion of Silica-Based Optical Fibers in Various Environments
Corros. Mater. Degrad. 2023, 4(3), 445-465; https://doi.org/10.3390/cmd4030023 - 08 Aug 2023
Abstract
►▼
Show Figures
This research article explores the potential of optical fibers as sensors, highlighting their ability to measure various parameters such as temperature, pressure, stress, and radiation dose. The study focuses on investigating the material compatibility of optical fibers in challenging sensing environments like Gen
[...] Read more.
This research article explores the potential of optical fibers as sensors, highlighting their ability to measure various parameters such as temperature, pressure, stress, and radiation dose. The study focuses on investigating the material compatibility of optical fibers in challenging sensing environments like Gen II/II+ and advance nuclear reactors, as well as concentrated solar power (CSP) plants. Material compatibility tests were conducted to determine the feasibility of using fluorine and germanium optical fiber sensors in these environments. The study found that raw fibers were corrosion-resistant to lead bismuth eutectic at 600 °C, regardless of the coating. In molten salt environments, raw fibers were incompatible with FLiNaK but showed corrosion resistance to MgCl₂-NaCl-KCl. However, the survivability of raw fiber optics improved with a gold coating in FLiNaK. Raw fiber optics were found to be incompatible in high-temperature steam at 1200 °C and in a pressurized water reactor (PWR) at 300 °C.
Full article

Figure 1
Open AccessReview
Concrete Alkali–Aggregate-Reactivity-Induced Steel Reinforcement Corrosion
by
and
Corros. Mater. Degrad. 2023, 4(3), 428-444; https://doi.org/10.3390/cmd4030022 - 21 Jul 2023
Abstract
►▼
Show Figures
The alkali–aggregate reactivity (AAR) of concrete, long known for mass concrete, can also induce corrosion of steel in reinforced concrete structures. Several examples are given for which the origin of observed reinforcement corrosion and loss of concrete cover originally was attributed to chloride-induced
[...] Read more.
The alkali–aggregate reactivity (AAR) of concrete, long known for mass concrete, can also induce corrosion of steel in reinforced concrete structures. Several examples are given for which the origin of observed reinforcement corrosion and loss of concrete cover originally was attributed to chloride-induced or to carbonation-induced reinforcement corrosion. Critical reviews of these cases, using available information, suggest that, more likely, the observed crack patterns and concrete deterioration are the result of long-term AAR-induced concrete matrix expansion and loss of concrete strength and that these effects occurred prior to the eventual initiation of reinforcement corrosion. This proposition is supported by finite element and other stress analyses of various concrete–steel ensembles. They show that concrete expansion produces tensile stresses localised at and near exterior concrete surfaces or relative to the reinforcement. The locations of high-stress and -strain zones so produced correlate with field observations of long-term concrete cracking and delamination. The present interpretations highlight that AAR may be a significant contributor to initiation and subsequent long-term development of reinforcement corrosion in structurally reinforced concretes.
Full article
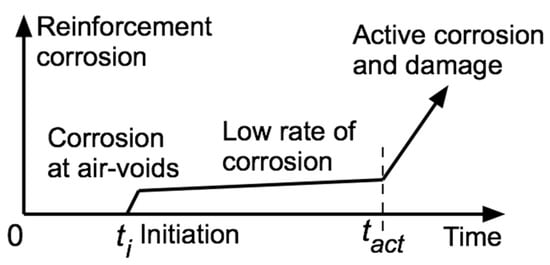
Figure 1
Open AccessFeature PaperArticle
Passive Oxide Destruction by Dense Low-Energy Radionuclide i-Analyzed by Voltammetry ii-Analyzed by Chaos
Corros. Mater. Degrad. 2023, 4(3), 398-427; https://doi.org/10.3390/cmd4030021 - 15 Jul 2023
Abstract
►▼
Show Figures
The destruction of the passive oxide can be caused by the action of a radionuclide, which collides with the surface of the oxide. In this case, the
The destruction of the passive oxide can be caused by the action of a radionuclide, which collides with the surface of the oxide. In this case, the

Figure 1
Open AccessFeature PaperArticle
Microbial Communities Associated with Alternative Fuels in Model Seawater-Compensated Fuel Ballast Tanks
by
, , , , and
Corros. Mater. Degrad. 2023, 4(3), 382-397; https://doi.org/10.3390/cmd4030020 - 03 Jul 2023
Cited by 1
Abstract
►▼
Show Figures
The biocorrosion of carbon steel poses a risk for ships combining seawater and fuel in metal ballast tanks. Ballast tanks were simulated by duplicate reactors containing carbon steel coupons and either petroleum F76 (petro-F76), Fischer–Tropsch F76 (FT-F76), or a 1:1 mix of both
[...] Read more.
The biocorrosion of carbon steel poses a risk for ships combining seawater and fuel in metal ballast tanks. Ballast tanks were simulated by duplicate reactors containing carbon steel coupons and either petroleum F76 (petro-F76), Fischer–Tropsch F76 (FT-F76), or a 1:1 mix of both fuels, to investigate whether the alternative fuel FT-F76 influenced this risk. The polycarbonate reactors were inoculated with seawater, and the control reactors did not receive fuel. The reactors were monitored for 400 days, and they all reached a pH and open circuit potential where elemental iron was oxidized, indicating corrosion. The reactors containing petro-76 or fuel mix had higher levels of dissolved iron; one of each replicate had lower concentrations of sulfate than the original seawater, while the sulfate concentration did not decrease in the other incubations. The high sulfate reactors, but not the low sulfate reactors, had a high relative abundance of microaerophilic sulfide-oxidizing bacteria. The FT-F76 and the no-fuel reactors had a high relative abundance of iron-sequestering Magnetovibrio. Although dissolved iron and loss of sulfate under anoxic conditions are associated with biocorrosion, our results suggest that in our reactors these indicators were altered by iron-sequestering and sulfide-oxidizing microbes, which is consistent with the slow diffusion of oxygen across the polycarbonate reactors.
Full article

Figure 1
Open AccessArticle
Investigation of the Effect Chloride Ions on Carbon Steel in Closed Environments at Different Temperatures
Corros. Mater. Degrad. 2023, 4(3), 364-381; https://doi.org/10.3390/cmd4030019 - 27 Jun 2023
Cited by 1
Abstract
►▼
Show Figures
This paper investigated carbon steel corrosion in an enclosed environment, where one set of steel was immersed in 3.5 wt.% NaCl solution and another exposed to humid (condensation) conditions. The study employed electrochemical noise and electrochemical impedance spectroscopy techniques to monitor real time
[...] Read more.
This paper investigated carbon steel corrosion in an enclosed environment, where one set of steel was immersed in 3.5 wt.% NaCl solution and another exposed to humid (condensation) conditions. The study employed electrochemical noise and electrochemical impedance spectroscopy techniques to monitor real time corrosion behaviour. The samples were evaluated with surface characterisation techniques including optical light microscope, scanning electron microscope with energy dispersive X-ray spectroscopy, and Raman spectroscopy. The results showed that carbon steel immersed in the liquid phase at a constant temperature exhibited the highest weight loss. However, the carbon steel exposed to water condensation (gas phase) at 80 °C manifested extensive localised corrosion. EN results agreed with the microscopy findings.
Full article
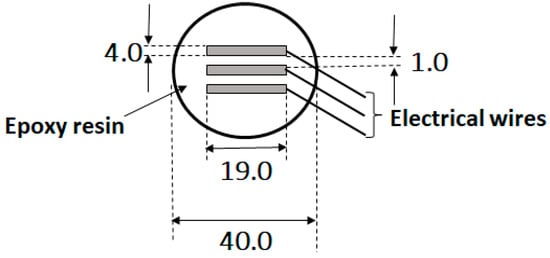
Figure 1
Open AccessArticle
Carbonation-Induced Corrosion of Reinforced Concrete Elements according to Their Positions in the Buildings
by
, , , , and
Corros. Mater. Degrad. 2023, 4(3), 345-363; https://doi.org/10.3390/cmd4030018 - 21 Jun 2023
Cited by 2
Abstract
►▼
Show Figures
Most regulations on the manufacturing of concrete for reinforced concrete structures rest on durability models that consider the corrosion of reinforcements. Those models are based on factors such as humidity, frost, presence of chlorides, and internal characteristics of the concrete itself, like resistance,
[...] Read more.
Most regulations on the manufacturing of concrete for reinforced concrete structures rest on durability models that consider the corrosion of reinforcements. Those models are based on factors such as humidity, frost, presence of chlorides, and internal characteristics of the concrete itself, like resistance, porosity, type of cement, water/cement ratio, etc. No regulations, however, adopt a purely constructive perspective when evaluating the risk of corrosion, i.e., the relative position of the reinforced concrete in buildings. The present work focuses on the relationship between the position of the damaged element and the building envelope. A total of 84 elements (columns and reinforced concrete beams) across twenty buildings were analysed in the provinces of Alicante and Murcia (Spain). The reinforcement concrete of these elements underwent carbonation-induced corrosion according to their positions in the buildings: (A) façade columns in contact with the ground; (B) interior columns in contact with the ground; (C) columns of walls in contact with the ground; (D) columns and external beams protected from rain; (E) columns and external beams exposed to rain; (F) columns and beams in air chambers under sanitary slabs; and (G), columns and interior beams. Of all types, elements (E) and (F) suffered carbonation-induced corrosion faster than the models used in the regulations, and type (G) underwent slower carbonation.
Full article
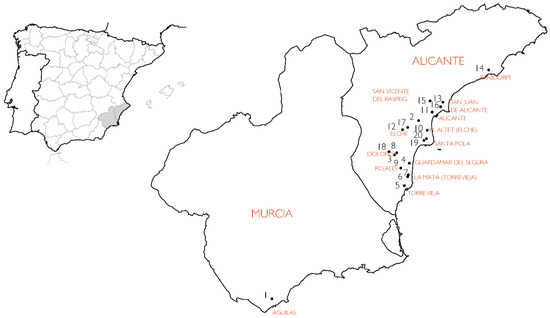
Figure 1
Open AccessArticle
Investigation on the Effect of a Chromium-Free Sealing Treatment for the Corrosion Resistance of AA2198-T851 after Tartaric Sulphuric Anodizing (TSA)
by
, , , and
Corros. Mater. Degrad. 2023, 4(2), 331-344; https://doi.org/10.3390/cmd4020017 - 12 Jun 2023
Abstract
►▼
Show Figures
The AA 2198-T851 is a third-generation Al-Li alloy developed for use in the aircraft industry. Al-Li alloys are susceptible to localized corrosion due to their complex microstructure resulting from the used thermomechanical treatment. In order to prevent localized corrosion, these alloys are usually
[...] Read more.
The AA 2198-T851 is a third-generation Al-Li alloy developed for use in the aircraft industry. Al-Li alloys are susceptible to localized corrosion due to their complex microstructure resulting from the used thermomechanical treatment. In order to prevent localized corrosion, these alloys are usually protected by anodizing in order to avoid a corrosive environment. Subsequently, for anodizing, a sealing treatment is usually performed for parts. Some sealing treatments use hexavalent-chromium-ion-containing solutions. In this investigation, a chromium-free sealing treatment in a solution with cerium ions has been carried out, and the effect on the corrosion resistance of the AA2198-T851 alloy was investigated. Hydrothermally sealed or unsealed samples were also tested for corrosion resistance for comparison reasons. The corrosion resistance of the anodized aluminum alloy, either hydrothermally sealed or in a cerium-ion-containing solution, was evaluated in a sodium chloride solution by electrochemical impedance spectroscopy as a function of immersion time. The samples sealed in a cerium-containing solution increased their corrosion resistance when compared to the hydrothermally sealed. The effectiveness of the sealing process with cerium that was observed in the electrochemical tests indicated that after the corrosive attack of the barrier layer, there was a “sealing” process of the sample surface.
Full article
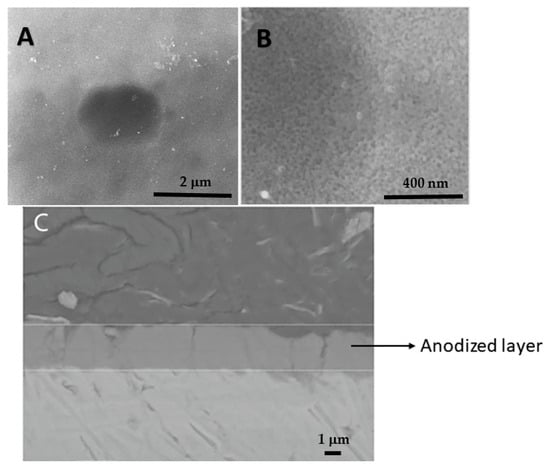
Figure 1
Open AccessArticle
Complete Desensitization of Aluminum–Magnesium Alloys via Boron Addition
Corros. Mater. Degrad. 2023, 4(2), 317-330; https://doi.org/10.3390/cmd4020016 - 06 Jun 2023
Abstract
►▼
Show Figures
We address here an important issue related to sensitization effects in Al5083 by mitigating the grain boundary precipitation of the beta phase and demonstrate that the addition of a small amount of boron to Al5083 impedes the precipitation of the beta phase, Al
[...] Read more.
We address here an important issue related to sensitization effects in Al5083 by mitigating the grain boundary precipitation of the beta phase and demonstrate that the addition of a small amount of boron to Al5083 impedes the precipitation of the beta phase, Al3Mg2, also known as the Samson phase. In Al–Mg alloys, the precipitation of Al3Mg2 usually occurs at grain boundaries in the temperature range of 50 to 200 °C from a supersaturated solid solution of Al–Mg and makes these alloys susceptible to intergranular corrosion and stress corrosion cracking. Upon boron addition, we show, using transmission electron microscopy, that a diboride phase, AlMgB2, forms at grain boundaries instead of the beta phase upon extended annealing at 150 °C. This diboride phase does not dissolve in saltwater, suggesting it is less anodic relative to the matrix. To quantify and compare the dissolution characteristics, we carried out nitric acid mass loss test for Al5083 samples containing 3 wt.% boron treated at 190 h at 150 °C, and fully sensitized Al5083 samples containing 0.0 wt.% boron. We estimate the mass loss to be 4 mg/cm2 for boron containing samples as compared to the mass loss of 45 mg/cm2 for samples without boron, indicating that the addition of boron is highly effective in suppressing the susceptibility to intergranular corrosion in Al5000 series alloys. This provides a potential route to minimize the longstanding problem of ship structure sensitization.
Full article

Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Coatings, CMD, Materials, Metals, Molecules
Corrosion and Protection of Metallic Materials, 2nd Edition
Topic Editors: Sebastian Feliú, Jr., Federico R. García-Galván, Lucien VelevaDeadline: 31 July 2024

Conferences
Special Issues
Special Issue in
CMD
Corrosion Mechanisms and Electrochemical Interfaces: In Honor of Prof. Digby Macdonald
Guest Editors: David M. Bastidas, Raman SinghDeadline: 31 July 2024









.jpg)





