Journal Description
Electrochem
Electrochem
is an international, peer-reviewed, open access journal on electrochemistry published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, CAPlus / SciFinder, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 22.3 days after submission; acceptance to publication is undertaken in 6.9 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
Latest Articles
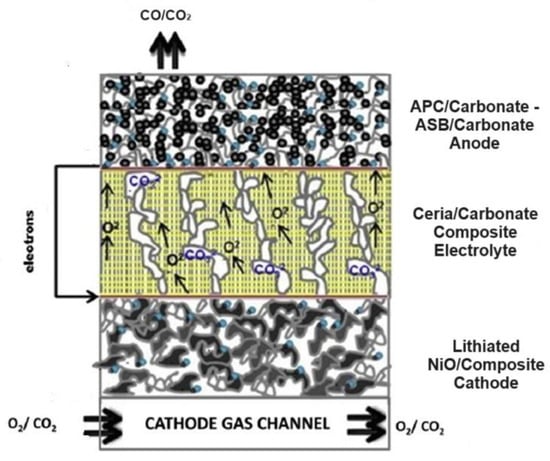
Artificial Intelligence for Electrochemical Prediction and Optimization of Direct Carbon Fuel Cells Fueled with Biochar
Electrochem 2024, 5(1), 29-44; https://doi.org/10.3390/electrochem5010002 - 04 Jan 2024
Abstract
At present, direct carbon fuel cells constitute an emerging energy technology that electrochemically converts solid carbon to electricity with high efficiency. The recent trend of DCFCs fueled with biochar from biomass carbonization as green fuel has reinforced the environmental benefits of DCFCs as
[...] Read more.
At present, direct carbon fuel cells constitute an emerging energy technology that electrochemically converts solid carbon to electricity with high efficiency. The recent trend of DCFCs fueled with biochar from biomass carbonization as green fuel has reinforced the environmental benefits of DCFCs as a clean and sustainable technology. However, there remain new challenges related to some complex unknown kinetic parameters,
(This article belongs to the Special Issue Advances in Electrochemical Energy Storage Systems)
►
Show Figures
Open AccessArticle
Identification of the Safe Variation Limits for the Optimization of the Measurements in Low-Cost Electrochemical Air Quality Sensors
Electrochem 2024, 5(1), 1-28; https://doi.org/10.3390/electrochem5010001 - 21 Dec 2023
Abstract
Nowadays, the study of air quality has become an increasingly prominent field of research, particularly in large urban centers, given its significant impact on human health. In many countries, government departments and research centers use official high-cost scientific instruments to monitor air quality
[...] Read more.
Nowadays, the study of air quality has become an increasingly prominent field of research, particularly in large urban centers, given its significant impact on human health. In many countries, government departments and research centers use official high-cost scientific instruments to monitor air quality in their regions. Meanwhile, concerned citizens interested in studying the air quality of their local areas often employ low-cost air quality sensors for monitoring purposes. The optimization and evaluation of low-cost sensors have been a field of research by many research groups. This paper presents an extensive study to identify the safe percentage change limits that low-cost electrochemical air quality sensors can have, in order to optimize their measurements. For this work, three low-cost air quality monitoring stations were used, which include an electrochemical sensor for nitrogen dioxide (NO2) (Alphasense NO2-B43F) and an electrochemical sensor for ozone (O3) (Alphasense OX-B431). The aim of this work is to explore the variance of the aforementioned sensors and how this variability can be used to optimize the measurements of low-cost electrochemical sensors, closer to real ones. The analysis is conducted by employing diagrams, boxplot and violin curves of the groups of sensors used, with satisfactory results.
Full article
(This article belongs to the Collection Feature Papers in Electrochemistry)
►▼
Show Figures

Figure 1
Open AccessReview
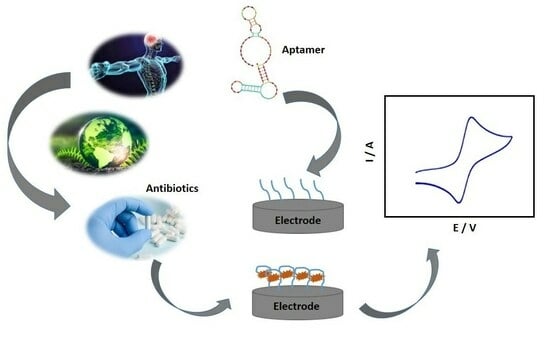
Relevant Aspects in the Development of Electrochemical Aptasensors for the Determination of Antibiotics—A Review
Electrochem 2023, 4(4), 553-567; https://doi.org/10.3390/electrochem4040035 - 12 Dec 2023
Abstract
Aptamers are three-dimensional structures of DNA or RNA that present high affinity and selectivity to specific targets, obtained through in vitro screening. Aptamers are used as biological recognizers in electrochemical biosensors, the so-called aptasensors, providing greater specificity in recognizing the most diverse analytes.
[...] Read more.
Aptamers are three-dimensional structures of DNA or RNA that present high affinity and selectivity to specific targets, obtained through in vitro screening. Aptamers are used as biological recognizers in electrochemical biosensors, the so-called aptasensors, providing greater specificity in recognizing the most diverse analytes. Electrochemical aptasensors have extremely relevant characteristics, such as high sensitivity, low cost compared to other biorecognizers such as antibodies, and excellent compatibility, being considered one of the most promising alternative methods in several areas, such as biomedical diagnosis and monitoring environmental contaminants. In this sense, the present work reviews the relevant aspects of methodologies based on electrochemical aptasensors and their applications in determining antibiotics, seeking to foster innovation in electrochemical biosensors.
Full article
(This article belongs to the Collection Feature Papers in Electrochemistry)
►▼
Show Figures
Graphical abstract
Open AccessReview
Electrochemical-Based Biosensor Platforms in Lab-Chip Models for Point-of-Need Toxicant Analysis
by
, , , , and
Electrochem 2023, 4(4), 537-552; https://doi.org/10.3390/electrochem4040034 - 21 Nov 2023
Abstract
►▼
Show Figures
The global hazardous waste management market is expected to reach USD 987.51 million by 2027 at a CAGR of 14.48%. The early detection of corrosive, flammable, and infectious toxicants from natural sources or manmade contaminants from different environments is crucial to ensure the
[...] Read more.
The global hazardous waste management market is expected to reach USD 987.51 million by 2027 at a CAGR of 14.48%. The early detection of corrosive, flammable, and infectious toxicants from natural sources or manmade contaminants from different environments is crucial to ensure the safety and security of the global living system. Even though the emergence of advanced science and technology continuously offers a more comfortable lifestyle, there are two sides of the coin in terms of opportunities and challenges, demanding solutions for greener applications and waste-to-wealth strategies. A modern analytical technique based on an electrochemical approach and microfluidics is one such emerging advanced solution for the early and effective detection of toxicants. This review attempts to highlight the different studies performed in the field of toxicant analysis, especially the fusion of electrochemistry and lab-chip model systems, promising for point-of-need analysis. The contents of this report are organised by classifying the types of toxicants and trends in electrochemical-integrated lab-chip assays that test for heavy-metal ions, food-borne pathogens, pesticides, physiological reactive oxygen/nitrogen species, and microbial metabolites. Future demands in toxicant analysis and possible suggestions in the field of microanalysis-mediated electrochemical (bio)sensing are summarised.
Full article
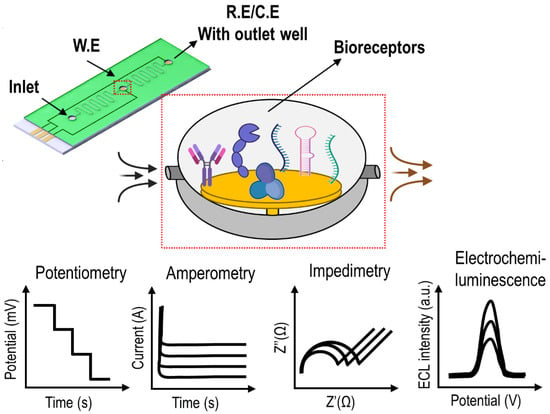
Figure 1
Open AccessArticle
A Disposable Carbon-Based Electrochemical Cell Modified with Carbon Black and Ag/δ-FeOOH for Non-Enzymatic H2O2 Electrochemical Sensing
by
, , , , , and
Electrochem 2023, 4(4), 523-536; https://doi.org/10.3390/electrochem4040033 - 14 Nov 2023
Abstract
►▼
Show Figures
Hydrogen peroxide (H2O2) is an essential analyte for detecting neurodegenerative diseases and inflammatory processes and plays a crucial role in pharmaceuticals, the food industry, and environmental monitoring. However, conventional H2O2 detection methods have drawbacks such as
[...] Read more.
Hydrogen peroxide (H2O2) is an essential analyte for detecting neurodegenerative diseases and inflammatory processes and plays a crucial role in pharmaceuticals, the food industry, and environmental monitoring. However, conventional H2O2 detection methods have drawbacks such as lengthy analysis times, high costs, and bulky equipment. Non-enzymatic sensors have emerged as promising alternatives to overcome these limitations. In this research, we introduce a simple, portable, and cost-effective non-enzymatic sensor that uses carbon black (CB) and silver nanoparticle-modified δ-FeOOH (Ag/δ-FeOOH) integrated into a disposable electrochemical cell (DCell). Scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDS), and electrochemical impedance spectroscopy (EIS) confirmed successful CB and Ag/δ-FeOOH immobilization on the DCell working electrode. Electrochemical investigations revealed that the DCell-CB//Ag/δ-FeOOH sensor exhibited an approximately twofold higher apparent heterogeneous electron transfer rate constant than the DCell-Ag/δ-FeOOH sensor, capitalizing on CB’s advantages. Moreover, the sensor displayed an excellent electrochemical response for H2O2 reduction, boasting a low detection limit of 22 µM and a high analytical sensitivity of 214 μA mM−1 cm−2. Notably, the DCell-CB//Ag/δ-FeOOH sensor exhibited outstanding selectivity for H2O2 detection, even in potential interferents such as dopamine, uric acid, and ascorbic acid. Furthermore, the sensor has the right qualities for monitoring H2O2 in complex biological samples, as evidenced by H2O2 recoveries ranging from 92% to 103% in 10% fetal bovine serum. These findings underscore the considerable potential of the DCell-CB//Ag/δ-FeOOH sensor for precise and reliable H2O2 monitoring in various biomedical and environmental applications.
Full article

Figure 1
Open AccessReview
Separator Materials for Lithium Sulfur Battery—A Review
by
Electrochem 2023, 4(4), 485-522; https://doi.org/10.3390/electrochem4040032 - 13 Nov 2023
Abstract
In the recent rechargeable battery industry, lithium sulfur batteries (LSBs) have demonstrated to be a promising candidate battery to serve as the next-generation secondary battery, owing to its enhanced theoretical specific energy, economy, and environmental friendliness. Its inferior cyclability, however, which is primarily
[...] Read more.
In the recent rechargeable battery industry, lithium sulfur batteries (LSBs) have demonstrated to be a promising candidate battery to serve as the next-generation secondary battery, owing to its enhanced theoretical specific energy, economy, and environmental friendliness. Its inferior cyclability, however, which is primarily due to electrode deterioration caused by the lithium polysulfide shuttle effect, is still a major problem for the real industrial usage of LSBs. The optimization of the separator and functional barrier layer is an effective strategy for remedying these issues. In this article, the current progress based on the classification and modification of functional separators is summarized. We will also describe their working mechanisms as well as the resulting LSB electrochemical properties. In addition, necessary performance for separators will also be mentioned in order to gain optimized LSB performance.
Full article
(This article belongs to the Topic Electrochemical Energy Storage Materials)
►▼
Show Figures
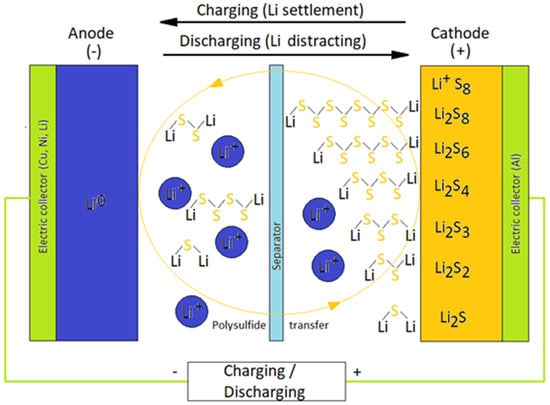
Figure 1
Open AccessArticle
Graphene-Oxide-Coated CoP2@C Anode Enables High Capacity of Lithium-Ion Batteries
by
, , , , , , , and
Electrochem 2023, 4(4), 473-484; https://doi.org/10.3390/electrochem4040031 - 26 Oct 2023
Cited by 1
Abstract
Cobalt diphosphides (CoP2) show a high theoretical capacity and hold great promise as anode materials for lithium-ion batteries (LIBs). However, the large variation in the volume and structure of CoP2 caused during lithium-ion insertion and extraction results in electrode fragmentation
[...] Read more.
Cobalt diphosphides (CoP2) show a high theoretical capacity and hold great promise as anode materials for lithium-ion batteries (LIBs). However, the large variation in the volume and structure of CoP2 caused during lithium-ion insertion and extraction results in electrode fragmentation and a compromised solid electrolyte interface, ultimately leading to poor cycling performance. Herein, a composite of CoP2 nanoparticles encapsulated in carbon matrix has been successfully synthesized by carbonization of Co-MOF-based zeolitic imidazolate frameworks (ZIF-67) and sequential phosphorization and further wrapped in graphene oxide (CoP2@C@GO). The formation of CoP2 was confirmed by X-ray diffraction, high-resolution transmission electron microscopy and X-ray photoelectron spectroscopy. The morphology of CoP2@C with and without GO wrapping was examined by scanning electron microscopy and transmission electron spectroscopy. It was demonstrated that the decoration of GO significantly reduces the polarization of CoP2@C electrodes, enhancing their charge capacity and cycling stability as an anode material for LIBs. After 200 cycles, they deliver a capacity of 450 mAh·g−1.
Full article
(This article belongs to the Collection Feature Papers in Electrochemistry)
►▼
Show Figures

Figure 1
Open AccessArticle
The Difference in the Effects of IR-Drop from the Negative Capacitance of Fast Cyclic Voltammograms
Electrochem 2023, 4(4), 460-472; https://doi.org/10.3390/electrochem4040030 - 23 Oct 2023
Cited by 2
Abstract
Diffusion-controlled cyclic voltammograms at fast scan rates show peak shifts, as well as decreases in the peak currents from predicted diffusion-controlled currents, especially when the currents are large in a low concentration of supporting electrolytes. This has been conventionally recognized as an IR
[...] Read more.
Diffusion-controlled cyclic voltammograms at fast scan rates show peak shifts, as well as decreases in the peak currents from predicted diffusion-controlled currents, especially when the currents are large in a low concentration of supporting electrolytes. This has been conventionally recognized as an IR-drop effect due to solution resistance on the peaks, as well as a heterogeneously kinetic effect. It is also brought about by the negatively capacitive currents associated with charge transfer reactions. The reaction product generates dipoles with counterions to yield a capacitance, the current of which flows oppositely to that of the double-layer capacitance. The three effects are specified here in the oxidation of a ferrocenyl derivative using fast scan voltammetry. The expression for voltammograms complicated with IR-drop is derived analytically and yields deformed voltammograms. The peak shift is approximately linear with the IR-voltage, but exhibits a convex variation. The dependence of some parameters on the peaks due to the IR-drop is compared with those due to the negative capacitance. The latter is more conspicuous than the former under conventional conditions. The two effects cannot be distinguished specifically except for variations in the conductance of the solution.
Full article
(This article belongs to the Special Issue Electrochemistry Modulated Interfacial Processes: Fundamental and Application)
►▼
Show Figures
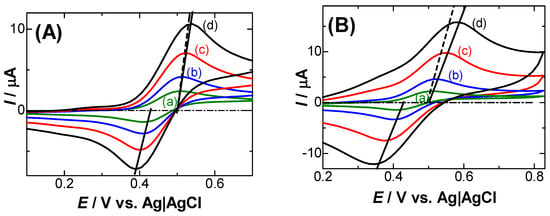
Figure 1
Open AccessArticle
New Analytical Expressions of Concentrations in Packed Bed Immobilized-Cell Electrochemical Photobioreactor
by
, , , and
Electrochem 2023, 4(4), 447-459; https://doi.org/10.3390/electrochem4040029 - 29 Sep 2023
Abstract
►▼
Show Figures
An electrochemical photobioreactor with a packed bed containing transparent gel granules and immobilized photosynthetic bacterial cells is shown with a one-dimensional two-phase flow and transport model. We consider the biological/chemical events in the electrochemical photobioreactor, the intrinsically connected two-phase flow and mass transport,
[...] Read more.
An electrochemical photobioreactor with a packed bed containing transparent gel granules and immobilized photosynthetic bacterial cells is shown with a one-dimensional two-phase flow and transport model. We consider the biological/chemical events in the electrochemical photobioreactor, the intrinsically connected two-phase flow and mass transport, and other factors. This model is based on a system of nonlinear equations. This paper applies Akbari-Ganji’s and Taylor series methods to find analytical solutions to nonlinear differential equations that arise in an immobilized-cell electrochemical photobioreactor. Approximate analytical expressions of the concentration of glucose and hydrogen are obtained in liquid and gas phases for different parameter values. Numerical simulations are presented to validate the theoretical investigations.
Full article
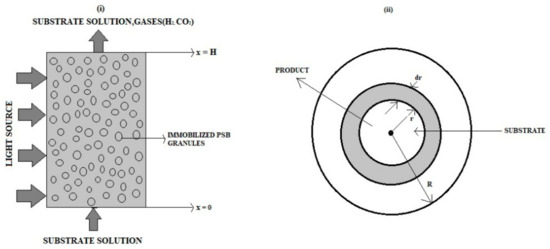
Figure 1
Open AccessArticle
Molecularly Imprinted Electrochemical Sensor Based on Poly (O-Phenylenediamine) for Sensitive Detection of Oxycodone in Water
Electrochem 2023, 4(4), 435-446; https://doi.org/10.3390/electrochem4040028 - 28 Sep 2023
Abstract
►▼
Show Figures
This work was aimed at the development of a sensitive electrochemical detection method for oxycodone in water. Molecularly imprinted electrodes were formed by electro-polymerization process using o-phenylenediamine as a monomer. The electro-polymerization was performed on glassy carbon electrodes in the presence of oxycodone
[...] Read more.
This work was aimed at the development of a sensitive electrochemical detection method for oxycodone in water. Molecularly imprinted electrodes were formed by electro-polymerization process using o-phenylenediamine as a monomer. The electro-polymerization was performed on glassy carbon electrodes in the presence of oxycodone before the extraction of entrapped oxycodone molecules. Various electrochemical techniques were employed to monitor the polymerization and response of the fabricated electrodes toward oxycodone. These techniques included cyclic voltammetry (CV), square wave voltammetry (SWV), differential pulse voltammetry (DPV) and electrochemical impedance spectroscopy (EIS). The oxycodone concentration was determined using SWV by measuring the change in the oxidation peak current of [Fe(CN)6]3−/4− in a 0.1 mM acetate buffer solution. At the optimal electro-polymerization conditions, a calibration curve of the current versus the concentration of oxycodone indicated a linear response at a region from 0.4 nM to 5.0 nM with a detection limit of 1.8 ± 0.239 nM. The MIP-modified electrode’s binding isotherm was fitted using a Langmuir model and showed an association constant, KA, of 1.12 × 106, indicating a high affinity of oxycodone molecules to binding sites. This sensor has the potential to act as an alternative method suitable for the on-site analysis of oxycodone.
Full article
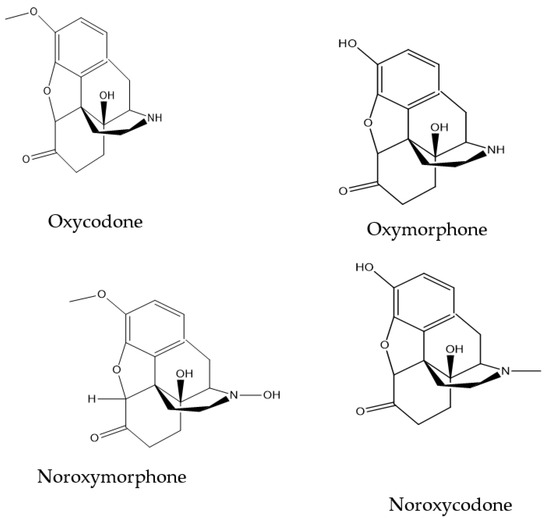
Figure 1
Open AccessArticle
Theory for Electrochemical Heat Sources and Exothermic Explosions: The Akbari–Ganji Method
Electrochem 2023, 4(3), 424-434; https://doi.org/10.3390/electrochem4030027 - 05 Sep 2023
Abstract
►▼
Show Figures
A device that transforms chemical energy into electrical energy is an electrochemical cell. The reaction type inside the cell determines whether it is exothermic or endothermic. This paper discusses the mathematical modelling of exothermic explosions in a slab. This model is based on
[...] Read more.
A device that transforms chemical energy into electrical energy is an electrochemical cell. The reaction type inside the cell determines whether it is exothermic or endothermic. This paper discusses the mathematical modelling of exothermic explosions in a slab. This model is based on a nonlinear equation containing a nonlinear term related to Arrhenius, bimolecular, and sensitised laws of reaction kinetics. The absolute temperature can be derived by solving the nonlinear equation using the Akbari–Ganji technique. The mathematical model also numerically solved and simulated in the MATLAB® v2016b software. The new simple theoretical result is validated with previously identified analytical and numerical findings. The influence of the parameters of Frank-Kamenetskii number, activation energy and the numerical exponent on temperature is discussed. The Frank-Kamenetskii number is observed to drop as the temperature is found to decrease, while the activation energy parameter is shown to increase. The numerical exponent has little or no effect on the temperature. An extension of this model to cylinder and sphere geometry is also provided.
Full article
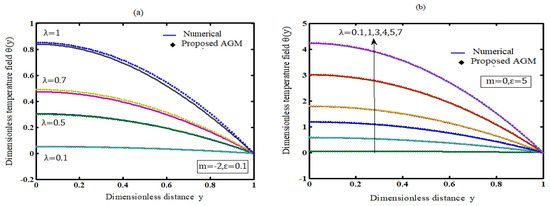
Figure 1
Open AccessReview
Design and Development of Food Waste Inspired Electrochemical Platform for Various Applications
by
Electrochem 2023, 4(3), 411-423; https://doi.org/10.3390/electrochem4030026 - 14 Aug 2023
Abstract
Plants have a remarkable position among renewable materials because of their abundance, and nearly thousands of tons are consumed worldwide every day. Most unexploited plants and agricultural waste can be a real potential resource system. With increasing environmental awareness and the growing importance
[...] Read more.
Plants have a remarkable position among renewable materials because of their abundance, and nearly thousands of tons are consumed worldwide every day. Most unexploited plants and agricultural waste can be a real potential resource system. With increasing environmental awareness and the growing importance of friendly agricultural waste, crops and fruit waste can be used for efficient conversion into bio-fertilizers, biocarbons, bio-polymers, biosensors and bio-fibers. Global challenges based on limited natural resources and fossil energy reserves simulated keen interest in the development of various electrochemical systems inspired by food and plant scraps, which aid in curbing pollution. The successful adoption of a renewable energy roadmap is dependent on the availability of a cheaper means of storage. In order to cut down the cost of storage units, an improvement on energy storage devices having better stability, power, and energy density with low post-maintenance cost is the vital key. Although food and plant scraps have a huge need for energy storage, it has been extended to various sensing platform fabrications, which are eco-friendly and comparable to organic molecule-based sensors. Current research proclivity has witnessed a huge surge in the development of phyto-chemical-based sensors. The state-of-the-art progresses on the subsequent use of plant-waste systems as nano-engineered electrochemical platforms for numerous environmental science and renewable energy applications. Moreover, the relevant rationale behind the use of waste in a well-developed, sustainable future device is also presented in this review.
Full article
(This article belongs to the Collection Feature Papers in Electrochemistry)
►▼
Show Figures
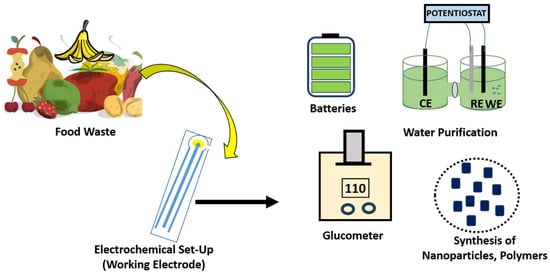
Figure 1
Open AccessArticle
Enabling Complex Impedance Spectroscopy for Cardio-Respiratory Monitoring with Wearable Biosensors: A Case Study
by
and
Electrochem 2023, 4(3), 389-410; https://doi.org/10.3390/electrochem4030025 - 10 Aug 2023
Abstract
►▼
Show Figures
Recent advances in commercially available integrated complex impedance spectroscopy controllers have brought rapid increases in the quality of systems available to researchers for wearable and remote patient monitoring applications. As a result, novel sensing methods and electrode configurations are increasingly viable, particularly for
[...] Read more.
Recent advances in commercially available integrated complex impedance spectroscopy controllers have brought rapid increases in the quality of systems available to researchers for wearable and remote patient monitoring applications. As a result, novel sensing methods and electrode configurations are increasingly viable, particularly for low-power embedded sensors and controllers for general electrochemical analysis. This study evaluates a case study of the four electrode locations suitable for wearable monitoring of respiratory and heart activity monitoring using complex impedance spectroscopy. We use tetrapolar electrode configurations with ten stimulation frequencies to characterize the relative differences in measurement sensitivity. Measurements are performed and compared for the magnitude, phase, resistive, and reactive components of the bioimpedance using two COTS-based controllers, the TI AFE4300 and MAX30009. We identify the highest percent relative changes in the magnitude of the impedance corresponding to deep breathing and heart activity across the chest (17% at 64 kHz, 0.5% at 256 kHz, respectively), on the forearm (0.098% at 16 kHz, 0.04% at 8 kHz), wrist-to-wrist across the body (0.28% at 256 kHz, 0.04% at 256 kHz, respectively), and wrist-to-finger across the body (0.35% at 4 kHz, 0.05% at 4 kHz, respectively). We demonstrate that the wrist-to-wrist and wrist-to-finger configurations are most promising and may enable new wearable bioimpedance applications. Additionally, this paper demonstrates that deep respiration and heart activity influence bioimpedance measurements in whole-body measurement configurations, with variations of nearly 1% in measured impedance due to the phase of the breathing cycle.
Full article
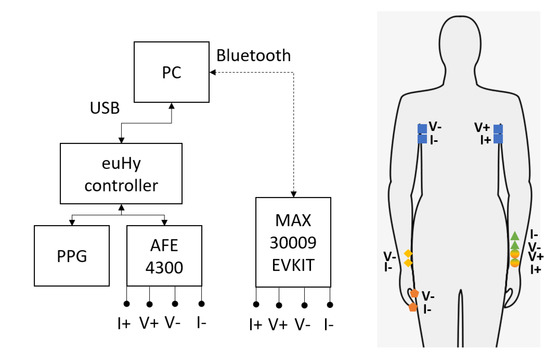
Figure 1
Open AccessReview
Emerging Trends in Nanomaterial-Based Biomedical Aspects
by
and
Electrochem 2023, 4(3), 365-388; https://doi.org/10.3390/electrochem4030024 - 04 Aug 2023
Abstract
►▼
Show Figures
Comprehending the interfacial interaction of nanomaterials (NMs) and biological systems is a significant research interest. NMs comprise various nanoparticles (NPs) like carbon nanotubes, graphene oxides, carbon dots, graphite nanopowders, etc. These NPs show a variety of interactions with biological interfaces via organic layers,
[...] Read more.
Comprehending the interfacial interaction of nanomaterials (NMs) and biological systems is a significant research interest. NMs comprise various nanoparticles (NPs) like carbon nanotubes, graphene oxides, carbon dots, graphite nanopowders, etc. These NPs show a variety of interactions with biological interfaces via organic layers, therapeutic molecules, proteins, DNA, and cellular matrices. A number of biophysical and colloidal forces act at the morphological surface to regulate the biological responses of bio-nanoconjugates, imparting distinct physical properties to the NMs. The design of future-generation nano-tools is primarily based on the basic properties of NMs, such as shape, size, compositional, functionality, etc., with studies being carried out extensively. Understanding their properties promotes research in the medical and biological sciences and improves their applicability in the health management sector. In this review article, in-depth and critical analysis of the theoretical and experimental aspects involving nanoscale material, which have inspired various biological systems, is the area of focus. The main analysis involves different self-assembled synthetic materials, bio-functionalized NMs, and their probing techniques. The present review article focuses on recent emerging trends in the synthesis and applications of nanomaterials with respect to various biomedical applications. This article provides value to the literature as it summarizes the state-of-the-art nanomaterials reported, especially within the health sector. It has been observed that nanomaterial applications in drug design, diagnosis, testing, and in the research arena, as well as many fatal disease conditions like cancer and sepsis, have explored alongwith drug therapies and other options for the delivery of nanomaterials. Even the day-to-day life of the synthesis and purification of these materials is changing to provide us with a simplified process. This review article can be useful in the research sector as a single platform wherein all types of nanomaterials for biomedical aspects can be understood in detail.
Full article

Figure 1
Open AccessArticle
Electrochemical Properties of Sputtered Ruthenium Oxide Neural Stimulation and Recording Electrodes
Electrochem 2023, 4(3), 350-364; https://doi.org/10.3390/electrochem4030023 - 24 Jul 2023
Abstract
A chronically stable electrode material with a low impedance for recording neural activity, and a high charge-injection capacity for functional electro-stimulation is desirable for the fabrication of implantable microelectrode arrays that aim to restore impaired or lost neurological functions in humans. For this
[...] Read more.
A chronically stable electrode material with a low impedance for recording neural activity, and a high charge-injection capacity for functional electro-stimulation is desirable for the fabrication of implantable microelectrode arrays that aim to restore impaired or lost neurological functions in humans. For this purpose, we have investigated the electrochemical properties of sputtered ruthenium oxide (RuOx) electrode coatings deposited on planar microelectrode arrays, using an inorganic model of interstitial fluid (model-ISF) at 37 °C as the electrolyte. Through a combination of cyclic voltammetry (CV) and an electrochemical impedance spectroscopy (EIS) modelling study, we have established the contribution of the faradaic reaction as the major charge-injection contributor within the safe neural stimulation potential window of ±0.6 V vs. Ag|AgCl. We have also established the reversibility of the charge-injection process for sputtered RuOx film, by applying constant charge-per-phase current stimulations at different pulse widths, and by comparing the magnitudes of the leading and trailing access voltages during voltage transient measurements. Finally, the impedance of the sputtered RuOx film was found to be reasonably comparable in both its oxidized and reduced states, although the electronic contribution from the capacitive double-layer was found to be slightly higher for the completely oxidized film around 0.6 V than for its reduced counterpart around −0.6 V.
Full article
(This article belongs to the Collection Feature Papers in Electrochemistry)
►▼
Show Figures

Figure 1
Open AccessReview
Use of Inner/Outer Sphere Terminology in Electrochemistry—A Hexacyanoferrate II/III Case Study
Electrochem 2023, 4(3), 313-349; https://doi.org/10.3390/electrochem4030022 - 11 Jul 2023
Cited by 3
Abstract
Salts of hexacyanoferrate II/III anions have been widely used as redox couple probe molecules to determine the characteristics of electrode surfaces. Examples include the assessment of electrocatalysts for energy applications and electrocatalysts for the detection of biological or chemical species, as well as
[...] Read more.
Salts of hexacyanoferrate II/III anions have been widely used as redox couple probe molecules to determine the characteristics of electrode surfaces. Examples include the assessment of electrocatalysts for energy applications and electrocatalysts for the detection of biological or chemical species, as well as the determination of electrochemically active surface areas. An examination of the electrochemical literature, based largely on cyclic voltammetric investigations, reveals a wide range of peak separation and/or heterogeneous electron transfer rate constants, classified sometimes as inner or outer sphere electron transfer processes. Originally developed for the mechanistic interpretation of inorganic transition metal compounds in solution, this terminology has since been extended to account for heterogeneous electron transfer occurring at electrodes. In the case of the hexacyanoferrate II/III anions, there can be a number of reasons why it sometimes behaves as an outer sphere probe and at other times displays inner sphere electron transfer characteristics. After examining some of the structural and chemical properties of the hexacyanoferrate II/III species, the methods used to determine such classifications are described. The most common method involves measuring peak-to-peak separation in a cyclic voltammogram to ascertain a heterogeneous rate constant, but it has inherent flaws. This paper reviews the reasons for the classification disparity, including the effects of various oxygen surface species, the influence of organic surface films, the nature of the cation counter-ion, surface adsorption and surface hydrophilicity/hydrophobicity. Other surface interactions may also take place, such as those occurring with Au corrosion or pH effects. These can impact the electrical double layer and thus may affect the electron transfer process. Consequently, it is recommended that hexacyanoferrate II/III should be considered a multi-sphere or alternatively a surface-sensitive electron transfer species.
Full article
(This article belongs to the Special Issue Electrochemistry Modulated Interfacial Processes: Fundamental and Application)
►▼
Show Figures
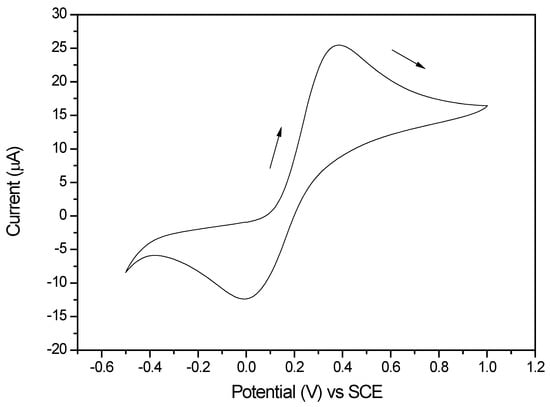
Figure 1
Open AccessArticle
Similarity of Heterogeneous Kinetics to Delay of Double-Layer Capacitance Using Chronoamperometry
Electrochem 2023, 4(2), 301-312; https://doi.org/10.3390/electrochem4020021 - 13 Jun 2023
Cited by 2
Abstract
Chronoamperometric curves for the oxidation of a ferrocenyl derivative via a potential step, calculated using the Cottrell equation, showed less diffusion-controlled currents on a platinum wire electrode. This lower deviation cannot be explained via Butler–Volmer heterogeneous kinetics, but was ascribed to the negatively
[...] Read more.
Chronoamperometric curves for the oxidation of a ferrocenyl derivative via a potential step, calculated using the Cottrell equation, showed less diffusion-controlled currents on a platinum wire electrode. This lower deviation cannot be explained via Butler–Volmer heterogeneous kinetics, but was ascribed to the negatively capacitive current associated with a redox reaction. The deviation in fully oxidized electrical potential corresponds to the non-zero concentration at the electrode surface, which cannot be predicted using the Nernst equation. This equation expresses the relationship between the electrical potential and activity at the electrode surface rather than the concentration. The diffusion equation determines the relationship between the current and surface concentration rather than activity. Negative capacitance or a non-zero concentration may arise from structure formation on the electrode owing to dipole–dipole interactions, which are similar to the generation of double-layer capacitance, including frequency dispersion. Following this concept, we derive expressions for a lowered diffusion-controlled current and time-dependent surface concentration. The negatively capacitive current shows the time dependence of t−0.9, which is similar to the decay of double-layer capacitive currents. The surface concentration decays with t−0.4-dependence.
Full article
(This article belongs to the Special Issue Electrochemistry Modulated Interfacial Processes: Fundamental and Application)
►▼
Show Figures
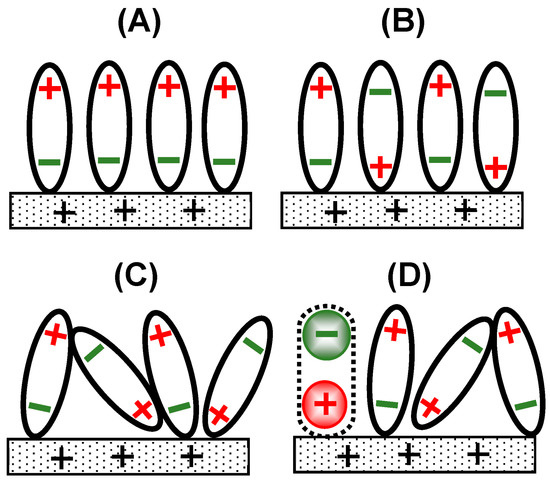
Figure 1
Open AccessArticle
Electrochemical Sensing of Amoxicillin Drug-Assisted Uropathogenic E. coli Bacteria Using Gold Nanostructures—A Preliminary Study
Electrochem 2023, 4(2), 291-300; https://doi.org/10.3390/electrochem4020020 - 13 Jun 2023
Abstract
The present study focuses on the electrochemical sensing of amoxicillin (AMX, as a model antibiotic drug) and its interaction with Uropathogenic E. coli (UPEC) bacteria (as a model pathogen) under physiological conditions. The electrochemical sensor probe is formulated by nanostructured gold wires (AuNWs)
[...] Read more.
The present study focuses on the electrochemical sensing of amoxicillin (AMX, as a model antibiotic drug) and its interaction with Uropathogenic E. coli (UPEC) bacteria (as a model pathogen) under physiological conditions. The electrochemical sensor probe is formulated by nanostructured gold wires (AuNWs) embedded in a carbon nanofiber–chitosan (CNF-CHIT) matrix. The synthesis of AuNWs is characterized by scanning electron microscopy (SEM), UV-Visible spectrophotometry, and X-ray photoelectron spectroscopy (XPS). The CNF-CHIT/AuNW-modified system is characterized by SEM and XPS. Initially, the CNF-CHIT/AuNW electrode was utilized for the sensing of AMX; later, in the antibiotic drug-assisted sensing of UPEC, i.e., in the presence of AMX, the interaction of UPEC was studied. The modified electrode showed appreciable sensitivity for AMX sensing; also, the interaction of AMX with UPEC is studied at two different conditions. One, at a fixed concentration of AMX (100 µM) and different concentrations of UPEC bacteria (0.6–1.2 × 106 CFU/mL), and another with incubation time (1 h–1 h 35 min) for bacterial reaction. The electrochemical antimicrobial resistance developed by UPEC, which is inherent in the sensing of AMX, is the key concept for the detection of pathogens.
Full article
(This article belongs to the Special Issue Electrochemistry Technologies in Bioanalysis and Electrochemical Immunosensor)
►▼
Show Figures
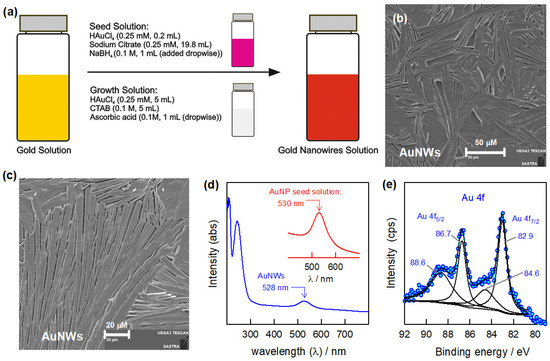
Figure 1
Open AccessReview
Current Updates in Bleomycin-Based Electrochemotherapy for Deep-Seated Soft-Tissue Tumors
by
, , , , , , and
Electrochem 2023, 4(2), 282-290; https://doi.org/10.3390/electrochem4020019 - 06 May 2023
Abstract
►▼
Show Figures
Electrochemotherapy (ECT) has evolved significantly during the last decade, expanding treatment indications from superficial skin lesions to advanced-stage, deep-seated tumors in hard-to-reach areas. Electrodes have also shown steady technological improvement throughout the years. Besides standard and VEG (variable geometry electrode) electrodes, the introduction
[...] Read more.
Electrochemotherapy (ECT) has evolved significantly during the last decade, expanding treatment indications from superficial skin lesions to advanced-stage, deep-seated tumors in hard-to-reach areas. Electrodes have also shown steady technological improvement throughout the years. Besides standard and VEG (variable geometry electrode) electrodes, the introduction of laparoscopic electrodes has brought on a new era in ECT treatment, making the minimally invasive approach a reality. The exact role of ECT in the oncological dashboard is yet to be determined; however, increased tumor response, pain relief, and a low number of adverse events may yield the way for more widespread application of the technique with possible further inclusion of ECT in international oncological guidelines. The aim of this review is to give an overview on the current status of ECT in deep-seated tumor treatment and shed light on its emerging role in local anticancer therapy.
Full article
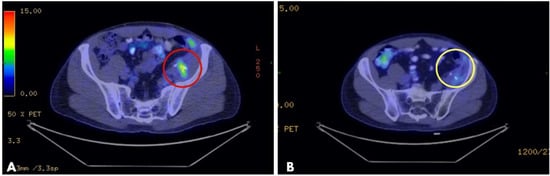
Figure 1
Open AccessCommunication
Electrochemical Oxidation Profile of Anthocyanin Keracyanin on Glassy and Screen-Printed Carbon Electrodes
Electrochem 2023, 4(2), 273-281; https://doi.org/10.3390/electrochem4020018 - 06 May 2023
Cited by 1
Abstract
A study of keracyanin chloride (KC) electrochemical behavior in an aqueous buffer solution using screen-printed carbon electrodes (SPCEs) and glassy carbon electrodes (GCEs) was performed. Cyclic voltammetry (CV) and square-wave voltammetry (SWV) were used to analyze the electrochemical response of KC under studied
[...] Read more.
A study of keracyanin chloride (KC) electrochemical behavior in an aqueous buffer solution using screen-printed carbon electrodes (SPCEs) and glassy carbon electrodes (GCEs) was performed. Cyclic voltammetry (CV) and square-wave voltammetry (SWV) were used to analyze the electrochemical response of KC under studied conditions. A clear redox wave was observed for KC, primarily due to the oxidation of the catechol 3′,4′-dihydroxyl group of its ring B, with a minor redox wave from oxidation of the hydroxyl groups in ring A. Compared to GCEs, using modified SPCEs resulted in two-fold amplification in the electrochemical oxidation signal of KC. Using SPCEs as a working electrode could provide high sensitivity in the quantification of KC and the ability to gauge KC quantification to significantly lower detection limits.
Full article
(This article belongs to the Collection Feature Papers in Electrochemistry)
►▼
Show Figures

Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Batteries, Electrochem, Materials, Nanomaterials, Molecules
Design and Mechanism of Aqueous Batteries
Topic Editors: Chuankun Zhang, Houzhao Wan, Pei Liang, Zhang LiDeadline: 30 April 2024
Topic in
Energies, Nanomaterials, Materials, Electrochem, Batteries
Electrochemical Energy Storage Materials
Topic Editors: Huang Zhang, Yuan MaDeadline: 30 June 2024
Topic in
Batteries, Electrochem, Energies, Materials, Polymers
Advances in Energy Storage Materials/Devices and Solid-State Batteries
Topic Editors: Claudio Gerbaldi, Federico Poli, Cataldo Simari, Akiko Tsurumaki, Francesca Soavi, Alessandro PiovanoDeadline: 31 August 2024

Conferences
Special Issues
Special Issue in
Electrochem
Silicon Electrochemistry: Fundamentals and Modern Applications
Guest Editor: Andrey SuzdaltsevDeadline: 7 July 2025