Journal Description
Future Pharmacology
Future Pharmacology
is an international, peer-reviewed, open access journal on pharmacology, drug discovery, and therapeutics published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 20.5 days after submission; acceptance to publication is undertaken in 6.6 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
- Future Pharmacology is a companion journal of Pharmaceutics.
Latest Articles
Hereditary Angioedema: Novel Molecules for Treatment of Acute Attacks and Long-Term Prophylaxis
Future Pharmacol. 2024, 4(1), 41-53; https://doi.org/10.3390/futurepharmacol4010005 - 12 Jan 2024
Abstract
►
Show Figures
Hereditary angioedema (HAE) is a rare disease caused by a genetic alteration of the SERPING1 gene and characterized by recurrent attacks of angioedema that involve the skin, and the mucosae of the gastrointestinal tract and upper airways, which significantly affect the quality of
[...] Read more.
Hereditary angioedema (HAE) is a rare disease caused by a genetic alteration of the SERPING1 gene and characterized by recurrent attacks of angioedema that involve the skin, and the mucosae of the gastrointestinal tract and upper airways, which significantly affect the quality of life of patients. Nowadays there are effective drugs for both 1. treating acute attacks and 2. preventing attacks with a long-term prophylaxis. However, there are some unmet needs for HAE treatment, and therefore several novel molecules are under active testing for this clinical condition. Novel drugs will simplify the mode of administration (oral versus parenteral for both on demand treatment or long-term prophylaxis), prolong the interval between administrations (up to 3–6 months of efficacy with a single administration), target more specifically the central enzymes involved in the generation of bradykinin, the ultimate mediator of angioedema (prekallikrein, activated plasma kallikrein or activated factor XII), and potentially determine a definitive cure for the disease by genetic manipulation of the altered gene (SERPING1) or other downstream genes (KLKB1). In this review we provide a panoramic view of all new medications that are under active experimentation and will probably transform and enrich all of the therapeutic armamentarium for treating this disease.
Full article
Open AccessArticle
Real-World Data Study on Risk Factors Associated with Acute Kidney Damage in Patients Treated with Anti-MRSA Antibiotics
by
, , , , , and
Future Pharmacol. 2024, 4(1), 30-40; https://doi.org/10.3390/futurepharmacol4010004 - 09 Jan 2024
Abstract
The objective was to evaluate the incidence of nephrotoxicity related to vancomycin and other anti-MRSA antibiotics (linezolid and daptomycin). Patients receiving any of these drugs between July 2014 and December 2020 at a tertiary hospital were included. Renal failure was evaluated using the
[...] Read more.
The objective was to evaluate the incidence of nephrotoxicity related to vancomycin and other anti-MRSA antibiotics (linezolid and daptomycin). Patients receiving any of these drugs between July 2014 and December 2020 at a tertiary hospital were included. Renal failure was evaluated using the acute renal injury (AKIN) system. Univariate analysis was conducted on the 5806 patients who were included. Among them, 1023 patients (17.62%) developed renal failure. The renal damage incidence was 14.74% (496/3365) for vancomycin, 19.13% (367/1918) for linezolid, and 30.59% (160/523) for daptomycin. Patients with lower basal glomerular filtration had a higher risk of AKIN. In the vancomycin group, the risk factors were high creatinine and urea serum basal values, duration of treatment (DOT), body mass index (BMI), ICU stay, age, and low CKDEPI and albumin levels. In the linezolid group, AKIN was linked to high creatinine and urea levels, BMI, age, and ICU stay and to low CKDEPI levels; for daptomycin, AKIN was associated with low CKDEPI and albumin levels and a long DOT. Patients with AKIN showed higher mortality rates. Vancomycin-associated nephrotoxicity remains a great concern. However, linezolid and daptomycin could also cause nephrotoxicity. Bearing in mind risk factors that may prompt nephrotoxicity in hospitalized patients taking anti-staphylococcal antibiotics will result in better pharmacotherapeutic management.
Full article
Open AccessArticle
The Dual Cardiovascular Effect of Centrally Administered Clonidine: A Comparative Study between Pentobarbital- and Ketamine/Xylazine-Anesthetized Rats
Future Pharmacol. 2024, 4(1), 17-29; https://doi.org/10.3390/futurepharmacol4010003 - 09 Jan 2024
Abstract
►▼
Show Figures
The administration of the α2-adrenergic receptor agonist clonidine via intracerebroventricular route produces hypotension in pentobarbital-anesthetized rats and pressor responses in conscious normotensive rats. We explored the impact of different anesthetics on the central nervous system-dependent cardiovascular effects of clonidine. Normotensive male
[...] Read more.
The administration of the α2-adrenergic receptor agonist clonidine via intracerebroventricular route produces hypotension in pentobarbital-anesthetized rats and pressor responses in conscious normotensive rats. We explored the impact of different anesthetics on the central nervous system-dependent cardiovascular effects of clonidine. Normotensive male Wistar rats with guide cannulas previously implanted in the cerebroventricular system were anesthetized with pentobarbital or ketamine/xylazine and prepared for blood pressure measurement. The animals received intracerebroventricular injections of 10 μg clonidine or 0.6 μg dexmedetomidine, and the effects on the systolic, diastolic, mean arterial pressure, and heart rate were evaluated. The influence of 5 μg yohimbine, a selective α2-adrenergic receptor antagonist, was also assessed. The i.c.v. microinjection of clonidine decreased all three components of systemic arterial pressure and the heart rate of pentobarbital-anesthetized rats. On the other hand, clonidine increased the blood pressure and generated a less intense reduction in the heart rate of ketamine/xylazine-anesthetized rats. The pressor and bradycardic effects of clonidine in ketamine/xylazine-anesthetized animals were reproduced by dexmedetomidine, a more selective α2-adrenergic receptor agonist. Notably, the previous intracerebroventricular injection of yohimbine significantly inhibited the hypertensive effect of clonidine and dexmedetomidine. This study discloses that while normotensive rats anesthetized with pentobarbital show hypotensive responses, the stimulation of α2-adrenergic receptors increases the blood pressure in rats under ketamine/xylazine-induced anesthesia, reproducing the effects seen in conscious normotensive animals. Recognizing the mechanisms involved in these differences may allow us to understand better the final effects of clonidine and other α2-adrenergic receptor agonists in the central nervous system, contributing to the repurposing of these drugs.
Full article
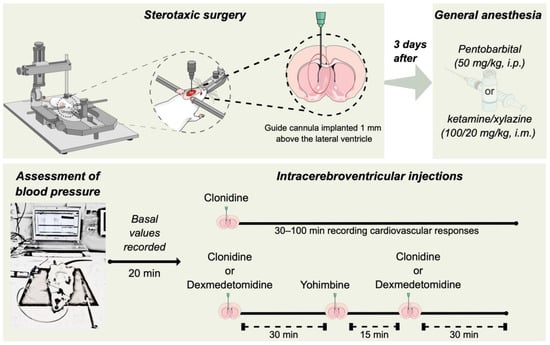
Figure 1
Open AccessArticle
Modulation of Staphylococcus aureus Biofilm Formation through Subinhibitory Concentrations of Biogenic Silver Nanoparticles and Simvastatin
by
, , , , and
Future Pharmacol. 2024, 4(1), 3-16; https://doi.org/10.3390/futurepharmacol4010002 - 05 Jan 2024
Abstract
Staphylococcus aureus is a causative agent of nosocomial infections and its antibiotic-resistant strains give cause for concern. Solutions are being explored to improve treatment for these infections, including repositioning drugs such as statins and using nanoparticles with antimicrobial properties. This study evaluated the
[...] Read more.
Staphylococcus aureus is a causative agent of nosocomial infections and its antibiotic-resistant strains give cause for concern. Solutions are being explored to improve treatment for these infections, including repositioning drugs such as statins and using nanoparticles with antimicrobial properties. This study evaluated the antimicrobial effects of simvastatin (SIM) and biologically synthesized silver nanoparticles (bio-AgNPs) in isolate form and in combination using assays of minimum inhibitory concentration (MIC), an in vitro biofilm model, and the association of antimicrobials against clinical strains of S. aureus. Bio-AgNPs showed a 53.8 ± 1.23 nm mean diameter and standard deviation, a 0.23 polydispersity index, and a −25.66 ± 2.19 mV mean potential and standard deviation. Transmission electron microscopy confirmed the formation of nanoparticles, and the presence of Ag0 and AgCl. S. aureus strains were sensitive to bio-AgNPs and SIM, showing 31.88–187.5 and 74.66–149.32 μM concentrations, respectively. The association assay showed 2.0 fractional inhibitory concentration indices (i.e., indifferent for clinical strains) and 0.32 values for the standard ATCC 29213 strain (synergy). Biofilm inhibition assays with isolated SIM and bio-AgNPs showed decreased biofilm formation 4× to ⅛ MICs concentrations, showing no synergism in association. These findings evince that simvastatin and bio-AgNPs at subinhibitory concentrations can serve as antimicrobial agents against S. aureus biofilm.
Full article
(This article belongs to the Special Issue Recent Advances in Pharmaceutical and Biomedical Analysis)
►▼
Show Figures
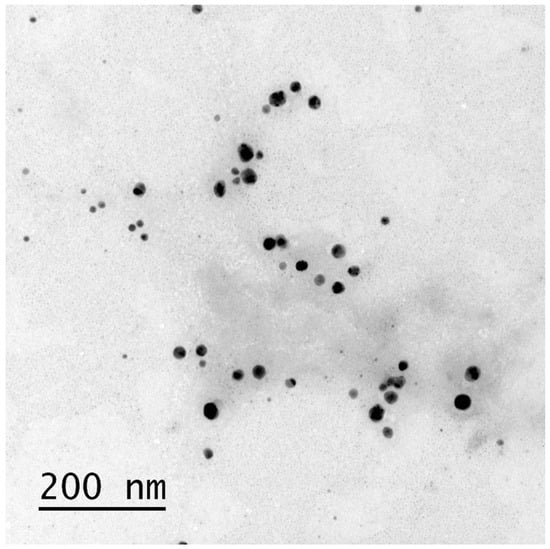
Figure 1
Open AccessEditorial
2023: The Best Year Ever for Future Pharmacology (and Even Better Years to Come)
Future Pharmacol. 2024, 4(1), 1-2; https://doi.org/10.3390/futurepharmacol4010001 - 02 Jan 2024
Abstract
The end of any year provides an opportunity to reflect on the past, and in particular, the past twelve months [...]
Full article
Open AccessReview
Therapeutic Use of Palmitoylethanolamide as an Anti-Inflammatory and Immunomodulator
Future Pharmacol. 2023, 3(4), 951-977; https://doi.org/10.3390/futurepharmacol3040058 - 15 Dec 2023
Abstract
Palmitoylethanolamine (PEA) is an endocannabinoid-like compound first encountered within the lipid fractions of specific foods and has intrigued researchers since the 1950s due to its therapeutic effects. This survey aims to explore the therapeutic promise held by PEA as an anti-inflammatory and immunomodulatory
[...] Read more.
Palmitoylethanolamine (PEA) is an endocannabinoid-like compound first encountered within the lipid fractions of specific foods and has intrigued researchers since the 1950s due to its therapeutic effects. This survey aims to explore the therapeutic promise held by PEA as an anti-inflammatory and immunomodulatory agent. The therapeutic impact of PEA reverberates across diverse physiological systems, such as the central nervous system, gastrointestinal tract, vascular network, and the digestive and respiratory system. Additionally, it is effective in pain management and reducing inflammation and immune responses. These attributes have fostered collaborations targeting conditions such as Alzheimer’s disease, multiple sclerosis, cerebral ischemia, neuroinflammation, general inflammation, pain, coagulopathy, steatohepatitis, and acute lung injury. PEA operates both independently and in synergy with other compounds, like paracetamol, luteolin, and oxymetazoline. This efficacy stems from its interactions with pivotal targets, including PPARα, PPAR-δ, PPAR-γ, CB1, CB2, GPR55, and TRPV1. Additionally, PEA exerts a direct influence on the inflammatory cascade, orchestrating precise adjustments in immune responses. Numerous animal studies have elucidated the inherent potential of PEA. Nevertheless, the imperative of reinforcing clinical investigation is evident. This review notably underscores the pivotal necessity for methodologically rigorous clinical trials to definitively establish the translational efficacy of PEA in ameliorating diverse inflammatory pathologies within the human milieu.
Full article
Open AccessReview
Gene-Based Therapy: A New Approach to Feline Induced Sterilization?
Future Pharmacol. 2023, 3(4), 938-950; https://doi.org/10.3390/futurepharmacol3040057 - 04 Dec 2023
Abstract
►▼
Show Figures
Feline population control remains a concern as to whether it is intended for the short- or long-term. Induced sterilization of felids is critical in the case of feral, free-roaming cats, or the management of wild populations in Zoos or sanctuaries. This narrative review
[...] Read more.
Feline population control remains a concern as to whether it is intended for the short- or long-term. Induced sterilization of felids is critical in the case of feral, free-roaming cats, or the management of wild populations in Zoos or sanctuaries. This narrative review explores the shifting paradigm in induced sterilization methods, driven by the development of gene editing approaches recently applied to control felid reproductive activity. Although gene therapy approaches have gained attention as alternatives to more traditional methods, their clinical applications remain in the realm of thought. The objective of this study was to provide a comprehensive overview of the current state and most recent advances in gene-based contraception options, consolidate current research and evidence, and share some considerations on its potential effectiveness, advantages or limitations, and implications for animal welfare and population control strategies. Gene-based contraception therapy tested in felines, targeting the AMH pathway, was unable to suppress the estrous cycle and follicular development. However, at an experimental level, preliminary results hint at the need to change towards different molecular targets. Moreover, their side effects remain largely unknown, and several questions remain unanswered, such as the regularity of treatment applications or cost.
Full article
Figure 1
Open AccessReview
Overview of Pharmacological Therapies for Diffuse Tenosynovial Giant Cell Tumor
by
, , , , , , and
Future Pharmacol. 2023, 3(4), 926-937; https://doi.org/10.3390/futurepharmacol3040056 - 01 Dec 2023
Abstract
►▼
Show Figures
Tenosynovial giant cell tumor (TGCT) is a rare and locally aggressive benign tumor arising from the synovium of joints, bursae, and tendon sheaths. It is classified into localized (L-TGCT) and diffuse (D-TGCT) forms based on the extent of involvement. Surgical resection is the
[...] Read more.
Tenosynovial giant cell tumor (TGCT) is a rare and locally aggressive benign tumor arising from the synovium of joints, bursae, and tendon sheaths. It is classified into localized (L-TGCT) and diffuse (D-TGCT) forms based on the extent of involvement. Surgical resection is the primary treatment, though achieving a definitive cure remains challenging due to the high recurrence rates, especially in D-TGCT. Systemic therapies targeting the CSF1-CSF1R axis have emerged as promising treatment options. CSF1R tyrosine kinase inhibitors (TKIs) such as imatinib, nilotinib, pexidartinib, and vimseltinib, alongside anti-CSF1R antibodies like emactuzumab, cabiralizumab, and lacnotuzumab, have shown encouraging results in managing TGCT, particularly when surgery is not feasible or poses significant morbidity. Other potential therapies, including local treatments and anti-inflammatory drugs, are being explored for TGCT management. This review provides an overview of systemic treatment options for D-TGCT, highlighting emerging therapeutic modalities and their potential implications. Effective management is crucial due to TGCT’s significant morbidity despite its non-life-threatening nature, necessitating novel approaches to improve patient prognosis and quality of life.
Full article

Figure 1
Open AccessArticle
Evaluations of NSAIDs and Opioids as Analgesics in Pediatric Oncology
by
, , , , , and
Future Pharmacol. 2023, 3(4), 916-925; https://doi.org/10.3390/futurepharmacol3040055 - 01 Dec 2023
Abstract
As one of the leading causes of death in childhood, cancer also causes discomfort to pediatric patients. Even with guidelines for pain management, more than half of hospitalized children have intense and unrelieved pain. The present work aims to describe the intensity of
[...] Read more.
As one of the leading causes of death in childhood, cancer also causes discomfort to pediatric patients. Even with guidelines for pain management, more than half of hospitalized children have intense and unrelieved pain. The present work aims to describe the intensity of pain and its pharmacological management in a pediatric oncology population. Patients aged 0 to 17 years old, diagnosed with cancer, who were admitted to a children’s oncology hospital and had well-documented data on pain management in their medical records were included. A total of 333 patients were included, mostly male (55.8%) with a mean age of 7.9 years. A substantial portion of the patient cohort (51.4%) initially reported experiencing pain of moderate intensity during the first assessment. Subsequently, following the pharmacological intervention, a significant proportion of patients (90.1%) reported complete alleviation of pain. The predominant pharmaceutical agents utilized for pain management encompassed metamizole (76.6%) and morphine (10.2%). All pharmacological interventions used were able to significantly reduce patients’ pain. This study underscores the utilization of different pharmacological classes to achieve notable reductions in pain intensity among patients grappling with severe pain.
Full article
Open AccessCase Report
Could Cariprazine Be a Possible Choice for High Functioning Autism? A Case Report
by
, , , , , , , and
Future Pharmacol. 2023, 3(4), 908-915; https://doi.org/10.3390/futurepharmacol3040054 - 01 Dec 2023
Abstract
This case report was conducted by searching for the following keywords on PubMed: High Functioning Autism, Autism Spectrum Disorder, cariprazine, aripiprazole, partial agonist antipsychotic, DRD2/DRD3. High Functioning Autism (HFA) is a neurodevelopmental disorder characterized by the core symptoms of autism spectrum disorder (ASD)
[...] Read more.
This case report was conducted by searching for the following keywords on PubMed: High Functioning Autism, Autism Spectrum Disorder, cariprazine, aripiprazole, partial agonist antipsychotic, DRD2/DRD3. High Functioning Autism (HFA) is a neurodevelopmental disorder characterized by the core symptoms of autism spectrum disorder (ASD) with average intellectual abilities, behavioral symptoms such as irritability, hyperactivity, aggressiveness and mood symptoms. HFA is not a term used in the Diagnostic and Statistical Manual of mental disorders (DSM), but it is commonly used to identify patients diagnosed with Autistic Disorder (AD) or Pervasive Developmental Disorder-Not Otherwise Specified (PDD-NOS) with average or above average intellectual abilities. Several factors are involved in HFA development, including environmental and genetic factors. In particular, over the last several decades, dopaminergic signaling system dysfunction has been highlighted as being responsible for behavioral patterns. Nowadays, symptoms of ASD lack a specific pharmacological treatment. The only medications approved by the Food and Drug Administration (FDA) for symptoms associated with ASD, in particular the irritability, are risperidone and aripiprazole. According to the hypothesis that dopamine receptor DRD2 and DRD3 might be involved in impulsive behavior, stereotypy, repetitive behaviors and language impairment, cariprazine could be a therapeutic option. This molecule is primarily characterized by DRD3 partial agonism and serotonin 5-HT1A partial agonism, with a lower ability to activate DRD2 than other third-generation antipsychotics, such as aripiprazole. We have reported here a case study of treatment of HFA with cariprazine.
Full article
Open AccessReview
Unlocking the Therapeutic Potential of Medicinal Plants for Alzheimer’s Disease: Preclinical to Clinical Trial Insights
Future Pharmacol. 2023, 3(4), 877-907; https://doi.org/10.3390/futurepharmacol3040053 - 13 Nov 2023
Abstract
►▼
Show Figures
Alzheimer’s disease (AD) is a progressive, multifactorial, and unremitting neurodegenerative disease characterized by memory loss, personality changes, and cognitive impairment. It has become more prevalent in recent years. Therefore, understanding the pathophysiology of AD and developing efficient therapeutic strategies are essential. Moreover, the
[...] Read more.
Alzheimer’s disease (AD) is a progressive, multifactorial, and unremitting neurodegenerative disease characterized by memory loss, personality changes, and cognitive impairment. It has become more prevalent in recent years. Therefore, understanding the pathophysiology of AD and developing efficient therapeutic strategies are essential. Moreover, the progression of the disease is unaffected by the pharmaceutical approaches discovered to date. Additionally, the failure of over 200 potential drug candidates in clinical trials over the past decade suggests the complexity and difficulty of both the disease and its underlying causes. Therefore, research focused on medicinal plant-based natural products in the search for novel neuroprotective therapeutic candidates for AD is essential. Indeed, several scientific investigations have demonstrated the efficacy of many medicinal plants and their principal phytochemicals in the treatment of AD. This review article covered the pathophysiological mechanisms of AD, the necessity for natural products as anti-AD treatments, and the most recent preclinical studies revealing the function of neuroprotective medicinal plants and their bioactive compounds in the effective management of AD. In addition, the review also presents clinical trial data of promising anti-AD formulations/agents of plant origin. Revealing recent findings and highlighting the clinical trial data related to the development of new treatments for AD would promote further research in this field and pave the way for the development of more effective and safe treatments for this debilitating disease.
Full article
Graphical abstract
Open AccessArticle
Sex-Specific Responses to Tacrolimus and Mycophenolate Mofetil in Spontaneously Hypertensive Rats
Future Pharmacol. 2023, 3(4), 862-876; https://doi.org/10.3390/futurepharmacol3040052 - 10 Nov 2023
Abstract
►▼
Show Figures
In recent decades, the roles of tacrolimus and mycophenolate mofetil (MMF) in hypertension have been under discussion. However, the question of whether there are sex-specific responses to these agents has not received enough attention. Aim: To evaluate sex-specific differences in the responses to
[...] Read more.
In recent decades, the roles of tacrolimus and mycophenolate mofetil (MMF) in hypertension have been under discussion. However, the question of whether there are sex-specific responses to these agents has not received enough attention. Aim: To evaluate sex-specific differences in the responses to tacrolimus and mycophenolate mofetil in female (F) and male (M) spontaneously hypertensive rats (SHRs) and evaluate whether T cells contribute to mean arterial pressure (MAP) changes. Methods: Male and female SHRs received either tacrolimus or MMF for 14 days. The rats were implanted with radiotelemeters. MAP was measured chronically; then, circulating and renal infiltrated CD4+, CD8+, T helper 17 (Th17), and T regulatory (Treg) cells were quantified using flow cytometry. Key Findings: Tacrolimus increased MAP only in males, and it decreased CD4+ and CD8+ T cells in both males and females (p < 0.05). The tacrolimus-induced reduction of renal CD4+ and Treg cells was more profound in males. MMF reduced MAP and circulating and renal CD4+ and CD8+ T cells in the male and female rats. MMF also decreased Th17 and Treg cells in both sexes, but the decrease in Th17 was higher in males (p < 0.05) and the reduction in Treg cells was higher in females (p < 0.05). Our findings indicate that the effects of tacrolimus and MMF on renal T cell subsets are sex-specific. Significance: Targeting T cells in hypertension using therapeutic agents may have different effects on men and women; so, the management of hypertension and post-transplant hypertension using these agents should be specified by gender.
Full article

Figure 1
Open AccessReview
Plasmodial Transcription Factors and Chromatin Modifiers as Drug Targets
by
, , , , , and
Future Pharmacol. 2023, 3(4), 846-861; https://doi.org/10.3390/futurepharmacol3040051 - 09 Nov 2023
Abstract
►▼
Show Figures
The complex life cycle of the malaria parasite Plasmodium requires the parasite to adequately adapt to different conditions. For this reason, Plasmodium strictly controls its gene expression, and given its evolutionary distance from the human host, the involved factors may figure as attractive
[...] Read more.
The complex life cycle of the malaria parasite Plasmodium requires the parasite to adequately adapt to different conditions. For this reason, Plasmodium strictly controls its gene expression, and given its evolutionary distance from the human host, the involved factors may figure as attractive potential drug targets. In recent years, several unique transcription factors and chromatin modifiers have been identified and partially characterized in Plasmodium falciparum and in the murine species P. yoelii and P. berghei. This review unites data from studies focusing on drug development against enigmatic plant-like AP2-transcription factors and chromatin modifiers, such as histone acetyl transferases and deacetylases and histone methyltransferases and demethylases. Considering the reported success of inhibition of both factors, these may be included as targets to effectively combat the parasite by perturbing its control of gene expression.
Full article

Figure 1
Open AccessReview
Considerations of Nystatin Roll in Oral Candidiasis Scenario and the COVID-19 Pandemic—A Review
by
, , and
Future Pharmacol. 2023, 3(4), 834-845; https://doi.org/10.3390/futurepharmacol3040050 - 07 Nov 2023
Abstract
►▼
Show Figures
Oral candidiasis is an opportunistic infection usually related to predisposing factors. Oral manifestations in patients affected by COVID-19 have been reported, as the oral mucosa is the gateway to this viral infection. Xerostomia, as well as other oral symptoms, are predisposing factors for
[...] Read more.
Oral candidiasis is an opportunistic infection usually related to predisposing factors. Oral manifestations in patients affected by COVID-19 have been reported, as the oral mucosa is the gateway to this viral infection. Xerostomia, as well as other oral symptoms, are predisposing factors for the emergence of oral candidiasis after the COVID-19 pandemic. It is a common pathology, but fatal if left untreated. Nystatin (NYS) is the drug of first choice in the treatment of oral candidiasis. Herein, we reviewed the epidemiology of oral candidiasis and its treatments, focusing on the mechanism of action, dosage forms, and NYS efficacy. NYS is an effective drug against oral candidiasis and belongs to Class IV of the biopharmaceutical classification system; however, its low solubility and low permeability may compromise its availability in the oral cavity and, consequently, its pharmacological action. Future perspectives to overcome drug limitations were also addressed and discussed in our review.
Full article

Figure 1
Open AccessArticle
Erythrocyte Folyl Polyglutamate Synthetase Activity Profiling as a Potential Tool for the Prediction of Methotrexate Efficacy and Toxicity in Rheumatoid Arthritis
Future Pharmacol. 2023, 3(4), 819-833; https://doi.org/10.3390/futurepharmacol3040049 - 06 Nov 2023
Abstract
►▼
Show Figures
Methotrexate (MTX) is the cornerstone of therapy in the treatment of rheumatoid arthritis (RA). However, its efficacy and toxicity are variable and remain unpredictable. Interindividual variation in the metabolism of MTX by the enzyme folyl polyglutamate synthetase (FPGS) has been associated with response
[...] Read more.
Methotrexate (MTX) is the cornerstone of therapy in the treatment of rheumatoid arthritis (RA). However, its efficacy and toxicity are variable and remain unpredictable. Interindividual variation in the metabolism of MTX by the enzyme folyl polyglutamate synthetase (FPGS) has been associated with response variability in RA. In this work, we propose the development of a FPGS phenotyping assay that can be evaluated as a tool for the prediction of efficacy and toxicity in patients with RA prior to initiating MTX therapy. FPGS activity was measured in erythrocyte lysate by monitoring methotrexate polyglutamate (MTX + Glun) formation using ultra-performance liquid chromatography tandem–mass spectrometry (UPLC/MS/MS). Erythrocyte FPGS activity was measured in newly diagnosed RA (n = 35) and osteoarthritis (n = 7) patients. The enzymatic assay was optimized for measuring FPGS activity in 25 µL of packed erythrocytes over two hours. The coefficient of variation for intra- and inter-day analysis was found to be 5% and 12%, respectively. The method was used to measure FPGS enzyme kinetics, resulting in a mean (SD) Km of 30.3 (4.8) µM and a Vmax of 612 (193) pmol MTX + Glu2/h/mL of packed erythrocytes. Mean (SD) erythrocyte FPGS activity in patients with RA was found to be 445.93 (344.50) pmol MTX + Glu2/h/mL and with a 26-fold difference in the range (range: 83–2179 pmol MTX + Glu2/h/mL) whereas for patients with OA, it was found to be 409.80 (157.66) pmol MTX + Glu2/h/mL with a 3.5-fold difference in the range (range: 200.95–683.93 pmol MTX + Glu2/h/mL). Monitoring erythrocyte FPGS activity may be a feasible strategy of phenotyping for methotrexate efficacy and toxicity in patients with RA.
Full article

Figure 1
Open AccessReview
Exploring Inflammasome Complex as a Therapeutic Approach in Inflammatory Diseases
by
, , , , , , and
Future Pharmacol. 2023, 3(4), 789-818; https://doi.org/10.3390/futurepharmacol3040048 - 02 Nov 2023
Abstract
►▼
Show Figures
Inflammasomes, a group of multiprotein complexes, are essential in regulating inflammation and immune responses. Several inflammasomes, including nucleotide-binding domain leucine-rich repeat-containing protein 1 (NLRP1), NLRP3, NLRP6, NLRP7, NLRP12, interferon-inducible protein 16 (IFI16), NOD-like receptor family CARD domain-containing protein 4 (NLRC4), absent in melanoma
[...] Read more.
Inflammasomes, a group of multiprotein complexes, are essential in regulating inflammation and immune responses. Several inflammasomes, including nucleotide-binding domain leucine-rich repeat-containing protein 1 (NLRP1), NLRP3, NLRP6, NLRP7, NLRP12, interferon-inducible protein 16 (IFI16), NOD-like receptor family CARD domain-containing protein 4 (NLRC4), absent in melanoma 2 (AIM2), and pyrin, have been studied in various inflammatory diseases. Activating inflammasomes leads to the processing and production of proinflammatory cytokines, such as interleukin (IL)-1β and IL-18. The NLRP3 inflammasome is the most extensively studied and well characterized. Consequently, targeting inflammasomes (particularly NLRP3) with several compounds, including small molecule inhibitors and natural compounds, has been studied as a potential therapeutic strategy. This review provides a comprehensive overview of different inflammasomes and their roles in six inflammatory diseases, including multiple sclerosis, Alzheimer’s disease, Parkinson’s disease, atherosclerosis, type 2 diabetes, and obesity. We also discussed different strategies that target inflammasomes to develop effective therapeutics.
Full article

Figure 1
Open AccessReview
Antimicrobial Peptides and Their Assemblies
Future Pharmacol. 2023, 3(4), 763-788; https://doi.org/10.3390/futurepharmacol3040047 - 19 Oct 2023
Abstract
►▼
Show Figures
Antibiotic resistance requires alternatives to fight multi-drug resistant strains. Antimicrobial peptides (AMPs) act by disrupting or solubilizing microbial cell walls or membranes in accordance with mechanisms difficult to counteract from the microbe’s point of view. In this review, structure–activity relationships for AMPs and
[...] Read more.
Antibiotic resistance requires alternatives to fight multi-drug resistant strains. Antimicrobial peptides (AMPs) act by disrupting or solubilizing microbial cell walls or membranes in accordance with mechanisms difficult to counteract from the microbe’s point of view. In this review, structure–activity relationships for AMPs and their assemblies are discussed, considering not only their self-assembly but also their interactions with their carriers for optimal delivery or their combinations with other complementary antimicrobials or moieties covalently bound to their chemical structure. The effect of the formulations on AMP activity is also evaluated, revealing a myriad of possibilities. Depending on the interaction forces between the AMP, the carrier, or the elements added to the formulations, AMP activity can be reduced, enhanced, or remain unaffected. Approaches protecting AMPs against proteolysis may also reduce their activity.
Full article

Figure 1
Open AccessReview
Carrier-Mediated Delivery of Low-Molecular-Weight N-Containing Drugs across the Blood–Brain Barrier or the Blood–Retinal Barrier Using the Proton-Coupled Organic Cation Antiporter
Future Pharmacol. 2023, 3(4), 742-762; https://doi.org/10.3390/futurepharmacol3040046 - 12 Oct 2023
Abstract
While it is true that pharmacotherapy has achieved desired health outcomes, significant unmet medical needs persist in the field of central nervous system (CNS) drugs, particularly for neurodegenerative diseases such as Alzheimer’s disease, as well as ocular diseases such as diabetic retinopathy and
[...] Read more.
While it is true that pharmacotherapy has achieved desired health outcomes, significant unmet medical needs persist in the field of central nervous system (CNS) drugs, particularly for neurodegenerative diseases such as Alzheimer’s disease, as well as ocular diseases such as diabetic retinopathy and age-related macular degeneration. Drugs cannot enter the brain from the bloodstream due to the presence of the blood–brain barrier (BBB). Similarly, they cannot enter the eyes from the bloodstream due to the blood–retina barrier (BRB), which is composed of the endothelium or the epithelium. Thus, innovative drug delivery systems that can overcome these barriers based on efflux transporters, hydrophobic lipid bilayer membranes, and tight junctions should be developed using patient-friendly techniques distinct from craniotomy procedures or intravitreal injections. Brain-penetrating CNS drugs and antihistamine drugs commonly share N-containing groups. These findings suggest that certain types of cation transporters are involved in their transportation across the cell membrane. Indeed, the proton-coupled organic cation (H+/OC) antiporter, whose specific characteristics remain unidentified, is responsible for transporting compounds with N-containing groups, such as clonidine and pyrilamine, at the BBB, and likely at the BRB as well. Therefore, well-designed low-molecular-weight drugs containing N-containing groups as transporter recognition units can enter the brain or the eyes through carrier-mediated transport. In this perspective review, I introduce the implementation and potential of H+/OC antiporter-mediated transport across the endothelium at the BBB or the BRB using drugs consciously designed with N-containing groups as their substrates.
Full article
(This article belongs to the Special Issue Unveiling New Insights and Treatment Options for Ocular Surface Diseases)
►▼
Show Figures

Figure 1
Open AccessArticle
Drug and Protein Interaction Network Construction for Drug Repurposing in Alzheimer’s Disease
by
, , , and
Future Pharmacol. 2023, 3(4), 731-741; https://doi.org/10.3390/futurepharmacol3040045 - 12 Oct 2023
Abstract
►▼
Show Figures
Alzheimer’s disease is one of the leading causes of death globally, significantly impacting countless families and communities. In parallel, recent advancements in molecular biology and network approaches, guided by the Network Medicine perspective, offer promising outcomes for Alzheimer’s disease research and treatment. In
[...] Read more.
Alzheimer’s disease is one of the leading causes of death globally, significantly impacting countless families and communities. In parallel, recent advancements in molecular biology and network approaches, guided by the Network Medicine perspective, offer promising outcomes for Alzheimer’s disease research and treatment. In this study, we aim to discover candidate therapies for AD through drug repurposing. We combined a protein-protein interaction (PPI) network with drug-target interactions. Experimentally validated PPI data were collected from the PICKLE meta-database, while drugs and their protein targets were sourced from the DrugBank database. Then, based on RNA-Seq data, we first assigned weights to edges to indicate co-expression, and secondly, estimated differential gene expression to select a subset of genes potentially related to the disease. Finally, small subgraphs (modules) were extracted from the graph, centered on the genes of interest. The analysis revealed that even if there is no drug targeting several genes of interest directly, an existing drug might target a neighboring node, thus indirectly affecting the aforementioned genes. Our approach offers a promising method for treating various diseases by repurposing existing drugs, thereby reducing the cost and time of experimental procedures and paving the way for more precise Network Medicine strategies.
Full article

Figure 1
Open AccessArticle
Green Tea Extract Solid Dispersion Pellets with Enhanced Permeability for Hyperlipidemia
by
, , , , and
Future Pharmacol. 2023, 3(4), 708-730; https://doi.org/10.3390/futurepharmacol3040044 - 10 Oct 2023
Abstract
►▼
Show Figures
Green tea extract, rich in polyphenols like catechins, has been reported to have pharmacological benefits in patients with hyperlipidemia. The minimal membrane permeability of green tea limits its use in terms of bioavailability. To improve the permeability of green tea catechins in order
[...] Read more.
Green tea extract, rich in polyphenols like catechins, has been reported to have pharmacological benefits in patients with hyperlipidemia. The minimal membrane permeability of green tea limits its use in terms of bioavailability. To improve the permeability of green tea catechins in order to enhance theiranti-hyperlipidemia activity, a surfactant-based polymer was used to formulate a solid dispersion of green tea and convert it into commercially acceptable pellets. Green tea extract solid dispersions (GTE-SDs) were prepared withsolvent evaporation method using Soluplus® as a carrier. The GTE-SDs were evaluated for ex vivo permeation studies and characterized using FTIR, DSC, and XRD for confirming the formation of SD. The GTE-SDs exhibiting enhanced ex vivo permeation of EGCG were converted into a pellet formulation using the extrusion spheronization technique while being optimized using a 32 full factorial design. Soluplus® exhibited a four-fold improvement in the ex vivo permeation of EGCG from GTE-SD pellets (33.27%) as compared to GTE (10.43%) (p-value < 0.0001). In male Wistar rats, optimized GTE-SD pellets reduced the lipid blood profiles as compared to GTE (p-value < 0.0001). Thus, GTE-SD pellets can serve as an effective drug delivery platform for hyperlipidemia.
Full article
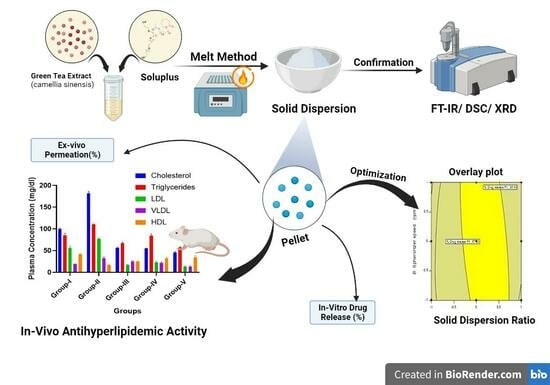
Graphical abstract
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Biomedicines, Cancers, Future Pharmacology, Pharmaceutics, Current Oncology
Recent Advances in Anticancer Strategies
Topic Editors: Hassan Bousbaa, Zhiwei HuDeadline: 30 June 2024

Conferences
Special Issues
Special Issue in
Future Pharmacology
Antiviral Drug Discovery and Development: Current Innovations and Future Trends
Guest Editor: Peng ZhanDeadline: 31 January 2024
Special Issue in
Future Pharmacology
Novel Therapeutic Approach to Inflammation and Pain
Guest Editors: Roberto Mattioli, Alessia MarianoDeadline: 31 March 2024
Special Issue in
Future Pharmacology
Unveiling New Insights and Treatment Options for Ocular Surface Diseases
Guest Editors: Paola Tirassa, Pamela Rosso, Fabiana MalloneDeadline: 30 April 2024
















