-
 Photothermal Imaging of Transient and Steady State Convection Dynamics in Primary Alkanes
Photothermal Imaging of Transient and Steady State Convection Dynamics in Primary Alkanes -
 Solvatochromic and Acid–Base Molecular Probes in Surfactant Micelles: Comparison of Molecular Dynamics Simulation with the Experiment
Solvatochromic and Acid–Base Molecular Probes in Surfactant Micelles: Comparison of Molecular Dynamics Simulation with the Experiment -
 Effect of Ligand Binding on Polymer Diffusiophoresis
Effect of Ligand Binding on Polymer Diffusiophoresis -
![Comprehensive Evaluation of End-Point Free Energy Techniques in Carboxylated-Pillar[6]arene Host-Guest Binding: IV. The QM Treatment, GB Models and the Multi-Trajectory Extension](https://pub.mdpi-res.com/title_story/title_story_17032268887149.jpg?1705329949) Comprehensive Evaluation of End-Point Free Energy Techniques in Carboxylated-Pillar[6]arene Host-Guest Binding: IV. The QM Treatment, GB Models and the Multi-Trajectory Extension
Comprehensive Evaluation of End-Point Free Energy Techniques in Carboxylated-Pillar[6]arene Host-Guest Binding: IV. The QM Treatment, GB Models and the Multi-Trajectory Extension
Journal Description
Liquids
Liquids
is an international, peer-reviewed, open access journal on all aspects of liquid material research published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within AGRIS, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 28 days after submission; acceptance to publication is undertaken in 4.9 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
Latest Articles
Polarity of Aqueous Solutions
Liquids 2024, 4(1), 107-116; https://doi.org/10.3390/liquids4010005 - 12 Jan 2024
Abstract
This short review describes the expansion of the solvatochromic approach utilizing water-soluble solvatochromic dyes to the analysis of solvent features of aqueous media in solutions of various compounds. These solvent features (polarity/dipolarity, hydrogen bond donor ability (HBD acidity), and hydrogen bond acceptor ability
[...] Read more.
This short review describes the expansion of the solvatochromic approach utilizing water-soluble solvatochromic dyes to the analysis of solvent features of aqueous media in solutions of various compounds. These solvent features (polarity/dipolarity, hydrogen bond donor ability (HBD acidity), and hydrogen bond acceptor ability (HBA basicity)) vary depending on the nature and concentration of a solute. Furthermore, the solvent features of water (the solvent dipolarity/polarizability and hydrogen bond donor ability) in solutions of various compounds describe multiple physicochemical properties of these solutions (such as the solubility of various compounds in aqueous solutions, salting-out and salting-in constants for polar organic compounds in the presence of different inorganic salts, as well as water activity, osmotic coefficients, surface tension, viscosity, and the relative permittivity of aqueous solutions of different individual compounds) and are likely related to changes in the arrangement of hydrogen bonds of water in these solutions.
Full article
(This article belongs to the Special Issue Solvatochromic Probes and Their Applications in Molecular Interaction Studies—a Themed Issue to Honor Professor Dr. Christian Reichardt)
►
Show Figures
Open AccessArticle
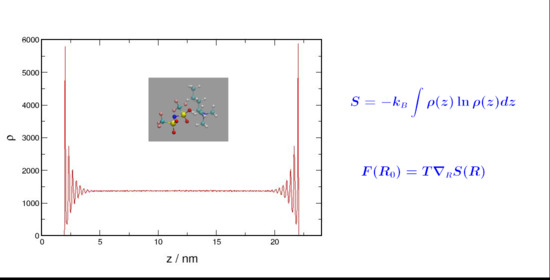
The Spatial Entropy of Confined Liquids
Liquids 2024, 4(1), 95-106; https://doi.org/10.3390/liquids4010004 - 08 Jan 2024
Abstract
►▼
Show Figures
Molecular dynamics simulations have been used to investigate the structural changes in confined liquids. The density distribution functions for weakly and strongly interacting liquids were determined and compared to those of a non-interacting system in order to assess the impact of the entropic
[...] Read more.
Molecular dynamics simulations have been used to investigate the structural changes in confined liquids. The density distribution functions for weakly and strongly interacting liquids were determined and compared to those of a non-interacting system in order to assess the impact of the entropic forces on the equilibrium state of the systems. The effect of the entropic forces was assessed by quantifying the layering on the liquid structure upon confinement. The more pronounced layering obtained for weakly interacting and non-interacting systems indicated that entropic forces are more effective in these systems where an increase in the multiplicity of states does not require a prohibitively high cost in energy.
Full article
Graphical abstract
Open AccessReview
Solvatochromism in Solvent Mixtures: A Practical Solution for a Complex Problem
Liquids 2024, 4(1), 73-94; https://doi.org/10.3390/liquids4010003 - 03 Jan 2024
Abstract
Many reactions are carried out in solvent mixtures, mainly because of practical reasons. For example, E2 eliminations are favored over SN2 substitutions in aqueous organic solvents because the bases are desolvated. This example raises the question: how do we chose binary
[...] Read more.
Many reactions are carried out in solvent mixtures, mainly because of practical reasons. For example, E2 eliminations are favored over SN2 substitutions in aqueous organic solvents because the bases are desolvated. This example raises the question: how do we chose binary solvents to favor reaction outcomes? This important question is deceptively simple because it requires that we understand the details of all interactions within the system. Solvatochromism (solvent-dependent color change of a substance) has contributed a great deal to answer this difficult question, because it gives information on the interactions between solvents, solute-solvent, and presumably transition state-solvent. This wealth of information is achieved by simple spectroscopic measurements of selected (solvatochromic) substances, or probes. An important outcome of solvatochromism is that the probe solvation layer composition is almost always different from that of bulk mixed solvent. In principle, this difference can be exploited to “tune” the composition of solvent mixture to favor the reaction outcome. This minireview addresses the use of solvatochromic probes to quantify solute-solvent interactions, leading to a better understanding of the complex effects of solvent mixtures on chemical phenomena. Because of their extensive use in chemistry, we focus on binary mixtures containing protic-, and protic-dipolar aprotic solvents.
Full article
(This article belongs to the Special Issue Solvatochromic Probes and Their Applications in Molecular Interaction Studies—a Themed Issue to Honor Professor Dr. Christian Reichardt)
►▼
Show Figures

Graphical abstract
Open AccessReview
Colloid Chemistry of Fullerene Solutions: Aggregation and Coagulation
Liquids 2024, 4(1), 32-72; https://doi.org/10.3390/liquids4010002 - 25 Dec 2023
Abstract
This review article is devoted to the colloidal properties of fullerene solutions. According to generally accepted understandings, all solvents in relations to fullerenes are divided into “good”, “poor”, and “reactive”. We have consistently considered the state of fullerenes in these systems. In “good”,
[...] Read more.
This review article is devoted to the colloidal properties of fullerene solutions. According to generally accepted understandings, all solvents in relations to fullerenes are divided into “good”, “poor”, and “reactive”. We have consistently considered the state of fullerenes in these systems. In “good”, predominantly non-polar aromatic solvents and CS2, non-equilibrium dissolution methods lead to the formation of colloidal aggregates, whereas the utilization of equilibrium methods results in the formation of molecular solutions. The latter, however, have some unusual properties; new results considered in this review confirm previously expressed ideas about colloidal properties of these solutions. In “poor” (polar) solvents, lyophobic colloidal systems appear. Both “bottom-up” and “top-down” methods of preparation are well documented in the literature. However, N-methylpyrrolidine-2-one, DMSO, and DMF dissolve fullerenes quite easily and with less energy consumption. These solvents can be considered a subset of “poor” solvents that have some features of being “reactive” at the expense of basic properties. New data confirm that hydrosols of fullerenes are typical hydrophobic colloids that obey the Schulze–Hardy rule and other regularities in the presence of electrolytes. Organosols in acetonitrile and methanol are much less stable with respect to the effects of electrolytes. This allows us to assume a non-DLVO stabilizing factor in the hydrosols. Accordingly, a new estimate of the Hamaker constant of fullerene–fullerene interaction is proposed. In DMSO and DMF, the coagulation of fullerene sols is hindered due to strong solvation with these basic solvents.
Full article
(This article belongs to the Special Issue Nanocarbon-Liquid Systems)
►▼
Show Figures
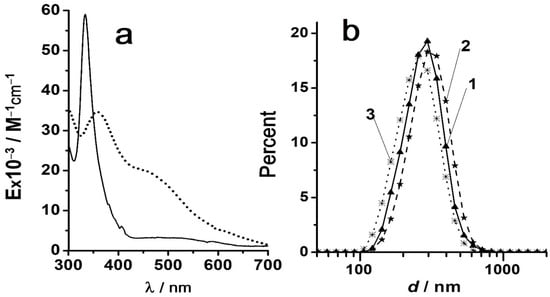
Figure 1
Open AccessReview
Inclusion Bodies in Ionic Liquids
Liquids 2024, 4(1), 1-31; https://doi.org/10.3390/liquids4010001 - 22 Dec 2023
Abstract
The pivotal role of proteins in pharmaceuticals is challenged by stability issues, making the study of inclusion bodies—a source of insoluble protein aggregates—increasingly relevant. This review outlines the critical procedures in inclusion body processing, focusing on ’mild solubilization concepts’ and refolding methodologies. Attention
[...] Read more.
The pivotal role of proteins in pharmaceuticals is challenged by stability issues, making the study of inclusion bodies—a source of insoluble protein aggregates—increasingly relevant. This review outlines the critical procedures in inclusion body processing, focusing on ’mild solubilization concepts’ and refolding methodologies. Attention is afforded to the emerging role of ionic liquids with unique and tunable physicochemical properties in optimizing protein unfolding and refolding processes. The review critically assesses the existing literature at the intersection of inclusion bodies and ionic liquids, identifying recent advancements, potential applications, and avenues for future research. This comprehensive analysis aims to elucidate the complexities in efficient protein processing from inclusion bodies.
Full article
(This article belongs to the Section Chemical Physics of Liquids)
►▼
Show Figures
Figure 1
Open AccessCommunication
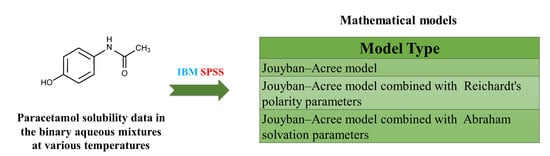
Prediction of Paracetamol Solubility in Binary Solvents Using Reichardt’s Polarity Parameter Combined Model
Liquids 2023, 3(4), 512-521; https://doi.org/10.3390/liquids3040032 - 14 Dec 2023
Abstract
The objective of this research is to propose a general model utilizing the solvatochromic polarity of electronic transition energy (ET) of the Reichardt indicator to predict paracetamol solubility in the solvent mixtures. In order to model validation, the available ET (30) values of
[...] Read more.
The objective of this research is to propose a general model utilizing the solvatochromic polarity of electronic transition energy (ET) of the Reichardt indicator to predict paracetamol solubility in the solvent mixtures. In order to model validation, the available ET (30) values of nine aqueous mixtures obtained from existing literature sources were utilized. The trained model yielded a relatively accurate estimation of paracetamol solubility in the investigated systems.
Full article
(This article belongs to the Special Issue Solvatochromic Probes and Their Applications in Molecular Interaction Studies—a Themed Issue to Honor Professor Dr. Christian Reichardt)
►▼
Show Figures
Graphical abstract
Open AccessReview
How the Concept of Solvent Polarity Investigated with Solvatochromic Probes Helps Studying Intermolecular Interactions
Liquids 2023, 3(4), 481-511; https://doi.org/10.3390/liquids3040031 - 06 Dec 2023
Abstract
Intermolecular interactions form the basis of the properties of solvents, such as their polarity, and are of central importance for chemistry; such interactions are widely discussed. Solvent effects were reported on the basis of various polarity probes with the ET(30) polarity
[...] Read more.
Intermolecular interactions form the basis of the properties of solvents, such as their polarity, and are of central importance for chemistry; such interactions are widely discussed. Solvent effects were reported on the basis of various polarity probes with the ET(30) polarity scale of Dimroth and Reichardt being of special interest because of its sensitivity, precise measurability and other advantages, and has been used for the investigation of solvent interactions. A two-parameter equation for the concentration dependence of medium effects has been developed, providing insights into structural changes in liquid phases. Moving from condensed gases to binary solvent mixtures, where the property of one solvent can be continuously transformed to the other, it was shown how the polarity of a solvent can be composed from the effect of polar functional groups and other structural elements that form the matrix. Thermochromism was discussed as well as the effect of very long-range interactions. Practical applications were demonstrated.
Full article
(This article belongs to the Special Issue Solvatochromic Probes and Their Applications in Molecular Interaction Studies—a Themed Issue to Honor Professor Dr. Christian Reichardt)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Revisiting the Total Hildebrand and Partial Hansen Solubility Parameters of Analgesic Drug Meloxicam
by
, , , and
Liquids 2023, 3(4), 469-480; https://doi.org/10.3390/liquids3040030 - 30 Nov 2023
Abstract
The reported total Hildebrand solubility parameter (δ2) value of meloxicam, as calculated based on the group contribution method proposed by Fedors, was compared with those estimated based on the maximum solubility peaks observed in different aqueous cosolvent systems at T
[...] Read more.
The reported total Hildebrand solubility parameter (δ2) value of meloxicam, as calculated based on the group contribution method proposed by Fedors, was compared with those estimated based on the maximum solubility peaks observed in different aqueous cosolvent systems at T = 298.15 K. Thus, the observed δ2 values varied from (19.8 to 29.1) MPa1/2, respectively. Moreover, the Hansen solubility parameters (HSPs) and the total Hildebrand solubility parameter were also determined by using the Bustamante regression method with the reported experimental solubility values of meloxicam in 31 neat solvents (30 organic solvents and water), obtaining the values: δd = 19.9 MPa1/2, δp = 16.9 MPa1/2, δh = 5.7 MPa1/2, and δT = 26.7 MPa1/2. Furthermore, the HSPs of meloxicam were also estimated based on the Hoftyzer–van Krevelen group contribution method, obtaining the values: δd = 17.9 MPa1/2, δp = 20.3 MPa1/2, and δh = 9.2 MPa1/2, and the total solubility parameter as: δT = 28.6 MPa1/2. In addition, the Kamlet–Abboud–Taft linear solvation energy relationship (KAT-LSER) model was also employed to evaluate the role of different intermolecular interactions on the dissolution of meloxicam in different solvents that varied in terms of polarity and hydrogen bonding capability.
Full article
(This article belongs to the Special Issue Solubility and Solubilization of Drugs: Modeling and Thermodynamic Analysis)
►▼
Show Figures

Figure 1
Open AccessReview
Oral Drug Delivery via Intestinal Lymphatic Transport Utilizing Lipid-Based Lyotropic Liquid Crystals
by
and
Liquids 2023, 3(4), 456-468; https://doi.org/10.3390/liquids3040029 - 20 Nov 2023
Abstract
Lyotropic liquid crystals (LLCs) are liquids that have crystalline structures. LLCs as drug delivery systems that can deliver hydrophobic, hydrophilic, and amphiphilic agents. Due to their unique phases and structures, LLCs can protect both small molecules and biologics from the gastrointestinal tract’s harsh
[...] Read more.
Lyotropic liquid crystals (LLCs) are liquids that have crystalline structures. LLCs as drug delivery systems that can deliver hydrophobic, hydrophilic, and amphiphilic agents. Due to their unique phases and structures, LLCs can protect both small molecules and biologics from the gastrointestinal tract’s harsh environment, thus making LLCs attractive as carriers for oral drug delivery. In this review, we discuss the advantages of LLCs and LLCs as oral formulations targeting intestinal lymphatic transport. In oral LLC formulations, the relationship between the micelle compositions and the resulting LLC structures as well as intestinal transport and absorption were determined. In addition, we further demonstrated approaches for the enhancement of intestinal lymphatic transport: (1) lipid-based LLCs promoting chylomicron secretion and (2) the design of LLC nanoparticles with M cell-triggered ligands for targeting the M cell pathway. In this review, we introduce LLC drug delivery systems and their characteristics. Our review focuses on recent approaches using oral LLC drug delivery strategies targeting the intestinal lymphatic system to enhance drug bioavailability.
Full article
(This article belongs to the Topic Recent Advances in Liquid Crystals)
►▼
Show Figures
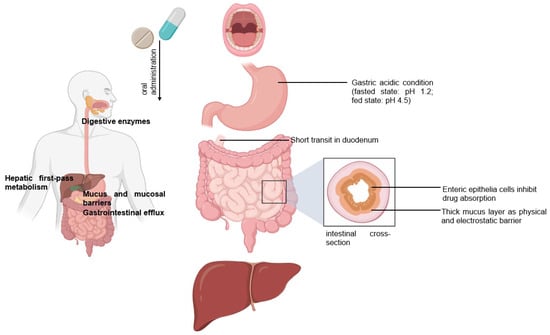
Figure 1
Open AccessArticle
Prediction of the Structural Color of Liquid Crystals via Machine Learning
by
, , , , , , , , , , , and
Liquids 2023, 3(4), 440-455; https://doi.org/10.3390/liquids3040028 - 15 Nov 2023
Abstract
►▼
Show Figures
Materials that generate structural color may be promising alternatives to dyes and pigments due to their relative long-term stability and environmentally benign properties. Liquid crystal (LC) mixtures of cholesteryl esters demonstrate structural color due to light reflected from the helical structure of the
[...] Read more.
Materials that generate structural color may be promising alternatives to dyes and pigments due to their relative long-term stability and environmentally benign properties. Liquid crystal (LC) mixtures of cholesteryl esters demonstrate structural color due to light reflected from the helical structure of the self-assembled molecules. The apparent color depends on the pitch length of the liquid crystal. While a wide range of colors have been achieved with such LC formulations, the nature of the pitch–concentration relationship has been difficult to define. In this work, various machine learning approaches to predict the reflected wavelength, i.e., the position of the selective reflection band, based on LC composition are compared to a Scheffe cubic model. The neural network regression model had a higher root mean squared error (RMSE) than the Scheffe cubic model with improved predictions for formulations not included in the dataset. Decision tree regression provided the best overall performance with the lowest RMSE and predicted position of the selective reflection band within 0.8% of the measured values for LC formulations not included in the dataset. The predicted values using the decision tree were over two-fold more accurate than the Scheffe cubic model. These results demonstrate the utility of machine learning models for predicting physical properties of LC formulations.
Full article

Figure 1
Open AccessArticle
Comprehensive Evaluation of End-Point Free Energy Techniques in Carboxylated-Pillar[6]arene Host-Guest Binding: IV. The QM Treatment, GB Models and the Multi-Trajectory Extension
Liquids 2023, 3(4), 426-439; https://doi.org/10.3390/liquids3040027 - 24 Oct 2023
Abstract
►▼
Show Figures
Due to the similarity of host–guest complexes and protein–ligand and protein–protein assemblies, computational tools for protein–drug complexes are commonly applied in host–guest binding. One of the methods with the highest popularity is the end-point free energy technique, which estimates the binding affinity with
[...] Read more.
Due to the similarity of host–guest complexes and protein–ligand and protein–protein assemblies, computational tools for protein–drug complexes are commonly applied in host–guest binding. One of the methods with the highest popularity is the end-point free energy technique, which estimates the binding affinity with gas-phase and solvation contributions extracted from simplified end-point sampling. Our series papers on a set of carboxylated-pillararene host–guest complexes have proven with solid numerical evidence that standard end-point techniques are practically useless in host–guest binding, but alterations, such as slightly increasing interior dielectric constant in post-processing calculation and shifting to the multi-trajectory realization in conformational sampling, could better the situation and pull the end-point method back to the pool of usable tools. Also, the force-field selection plays a critical role, as it determines the sampled region in the conformational space. In the current work, we continue the efforts to explore potentially promising end-point modifications in host–guest binding and further extend the sampling time to an unprecedent length. Specifically, we comprehensively benchmarked the shift from the original MM description to QM Hamiltonians in post-processing the popular single-trajectory sampling. Two critical settings in the multi-scale QM/GBSA regime are the selections of the QM Hamiltonian and the implicit-solvent model, and a scan of combinations of popular semi-empirical QM Hamiltonians and GB models is performed. The multi-scale QM/GBSA treatment is further combined with the three-trajectory sampling protocol, introducing a further advanced modification. The sampling lengths in the host–guest complex, solvated guest and solvated host ensembles are extended to 500 ns, 500 ns and 12,000 ns. As a result, the sampling quality in end-point calculations is unprecedently high, enabling us to draw conclusive pictures of investigated forms of modified end-point free energy methods. Numerical results suggest that the shift to the QM Hamiltonian does not better the situation in the popular single-trajectory regime, but noticeable improvements are observed in the three-trajectory sampling regime, especially for the DFTB/GBSA parameter combination (either DFTB2 or its third-order extension), the quality metrics of which reach an unprecedently high level and surpass existing predictions (including costly alchemical transformations) on this dataset, hinting on the applicability of the advanced three-trajectory QM/GBSA end-point modification for host–guest complexes.
Full article

Figure 1
Open AccessArticle
Effect of Ligand Binding on Polymer Diffusiophoresis
Liquids 2023, 3(4), 414-425; https://doi.org/10.3390/liquids3040026 - 19 Oct 2023
Abstract
►▼
Show Figures
Diffusiophoresis is the migration of a macromolecule in response to a concentration gradient of a cosolute in liquids. Diffusiophoresis of polyethylene glycol (PEG) in water occurs from high to low concentration of trimethylamine-N-oxide (TMAO). This is consistent with the preferential hydration of PEG
[...] Read more.
Diffusiophoresis is the migration of a macromolecule in response to a concentration gradient of a cosolute in liquids. Diffusiophoresis of polyethylene glycol (PEG) in water occurs from high to low concentration of trimethylamine-N-oxide (TMAO). This is consistent with the preferential hydration of PEG observed in the presence of TMAO. In other words, PEG migrates in the direction in which it lowers its chemical potential. On the other hand, although PEG is found to preferentially bind urea in water, PEG diffusiophoresis still occurs from high to low urea concentration. Thus, PEG migrates in the direction that increases its chemical potential in the urea case. Here, a ligand-binding model for polymer diffusiophoresis in the presence of a cosolute that preferentially binds to polymer is developed. It includes both actual polymer–ligand binding and the effect of the polymer on cosolute thermodynamic activity. This model shows that polymer–cosolute binding has a marginal effect on polymer diffusiophoresis and indicates that weak repulsive interactions, such as hard-core exclusion forces, are the main factor responsible for the observed PEG diffusiophoresis from high to low urea concentration. This work contributes to a better understanding of diffusiophoresis of macromolecules in response to gradients of nonelectrolytes.
Full article

Figure 1
Open AccessArticle
Abraham Model Descriptors for Vitamin K4: Prediction of Solution, Biological and Thermodynamic Properties
Liquids 2023, 3(4), 402-413; https://doi.org/10.3390/liquids3040025 - 02 Oct 2023
Cited by 1
Abstract
Spectrophotometric measurements were used to determine the mole fraction solubilities of vitamin K4 dissolved in cyclohexane, methylcyclohexane, 1-heptanol, 2-butanol, 2-pentanol, 2-methyl-1-butanol, 4-methyl-2-pentanol, and cyclopentanol at 298.15 K. Results from our experimental measurements, combined with the published solubility data, are used to calculate the
[...] Read more.
Spectrophotometric measurements were used to determine the mole fraction solubilities of vitamin K4 dissolved in cyclohexane, methylcyclohexane, 1-heptanol, 2-butanol, 2-pentanol, 2-methyl-1-butanol, 4-methyl-2-pentanol, and cyclopentanol at 298.15 K. Results from our experimental measurements, combined with the published solubility data, are used to calculate the solute descriptors of the vitamin K4 solute. The calculated solute descriptors describe the observed solubility data to within an overall standard deviation of 0.110 log units. The calculated solute descriptors were also used to estimate the several blood-to-rat tissue partition coefficients of vitamin K4, as well as the equilibrium vapor pressure above the solid vitamin at 298 K, and the vitamin’s enthalpy of solvation in both water and in 1,4-dioxane organic mono-solvent.
Full article
(This article belongs to the Special Issue Solubility and Solubilization of Drugs: Modeling and Thermodynamic Analysis)
►▼
Show Figures

Figure 1
Open AccessCommunication
Effective Recognition of Lithium Salt in (Choline Chloride: Glycerol) Deep Eutectic Solvent by Reichardt’s Betaine Dye 33
Liquids 2023, 3(4), 393-401; https://doi.org/10.3390/liquids3040024 - 28 Sep 2023
Abstract
Deep eutectic solvents (DESs) have emerged as novel alternatives to common solvents and VOCs. Their employment as electrolytes in batteries has been an area of intense research. In this context, understanding changes in the physicochemical properties of DESs in the presence of Li
[...] Read more.
Deep eutectic solvents (DESs) have emerged as novel alternatives to common solvents and VOCs. Their employment as electrolytes in batteries has been an area of intense research. In this context, understanding changes in the physicochemical properties of DESs in the presence of Li salts becomes of utmost importance. Solvatochromic probes have the potential to gauge such changes. It is reported herein that one such UV–vis molecular absorbance probe, Reichardt’s betaine dye 33, effectively manifests changes taking place in a DES Glyceline composed of H-bond accepting salt choline chloride and H-bond donor glycerol in a 1:2 molar ratio, as salt LiCl is added. The lowest energy intramolecular charge–transfer absorbance band of this dye exhibits a 17 nm hypsochromic shift as up to 3.0 molal LiCl is added to Glyceline. The estimated
(This article belongs to the Special Issue Solvatochromic Probes and Their Applications in Molecular Interaction Studies—a Themed Issue to Honor Professor Dr. Christian Reichardt)
►▼
Show Figures
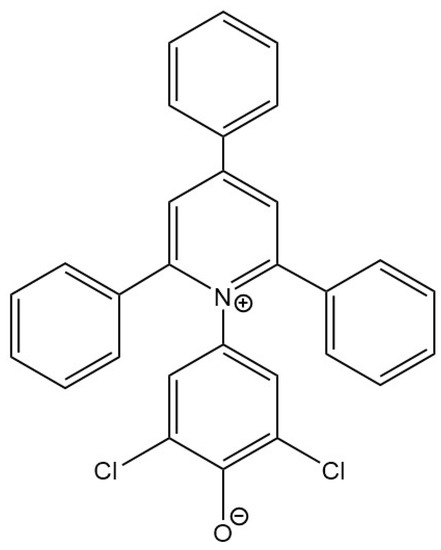
Figure 1
Open AccessCommunication
Behavior of C70 Fullerene in a Binary Mixture of Xylene and Tetrahydrofuran
by
, , , , and
Liquids 2023, 3(3), 385-392; https://doi.org/10.3390/liquids3030023 - 06 Sep 2023
Cited by 1
Abstract
The self-organization properties of C70 fullerene molecules in a xylene/tetrahydrofuran binary mixture were studied for the first time by optical absorption, refractometry, and dynamic light scattering. A correlation has been established between the change in the refractive index of the C70
[...] Read more.
The self-organization properties of C70 fullerene molecules in a xylene/tetrahydrofuran binary mixture were studied for the first time by optical absorption, refractometry, and dynamic light scattering. A correlation has been established between the change in the refractive index of the C70/xylene/tetrahydrofuran solution and the degree of self-organization of C70 molecules in the medium at various concentrations and storage periods of the solution. It is shown that the features of the optical absorption spectrum of C70/xylene/tetrahydrofuran at a fixed low concentration of fullerene are sensitive to its storage time. It was determined that the beginning time of the formation of C70 nanoclusters and their final size depend on the degree of concentration of fullerene and the time spent keeping the solution. The observed nature of the C70 fullerene solution in a binary mixture may help to elucidate its mechanism of self-organization in the future.
Full article
(This article belongs to the Special Issue Nanocarbon-Liquid Systems)
►▼
Show Figures
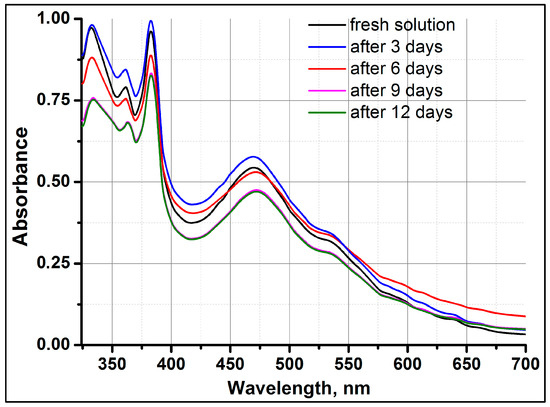
Figure 1
Open AccessArticle
Photothermal Imaging of Transient and Steady State Convection Dynamics in Primary Alkanes
Liquids 2023, 3(3), 371-384; https://doi.org/10.3390/liquids3030022 - 01 Sep 2023
Abstract
►▼
Show Figures
This paper presents a photothermal spectroscopy technique that effectively images convective heat flow in molecular liquids resulting from localized laser-induced heating. The method combines aspects of thermal lensing and photothermal deflection. A high-energy infrared laser is used to induce a thermal lens in
[...] Read more.
This paper presents a photothermal spectroscopy technique that effectively images convective heat flow in molecular liquids resulting from localized laser-induced heating. The method combines aspects of thermal lensing and photothermal deflection. A high-energy infrared laser is used to induce a thermal lens in the sample, and a divergent visible laser is used to probe the entire region of the excitation beam within the sample. This approach allows for the observation of the convective flow of the liquid above the excitation beam. The study focuses on the liquid primary alkanes, from n-pentane to n-pentadecane. The paper provides experimental results, including dynamical data for the propagation of the thermal plume, a transient feature, in these alkanes and the exploration of dependence on excitation laser power.
Full article

Figure 1
Open AccessReview
Solvatochromic and Acid–Base Molecular Probes in Surfactant Micelles: Comparison of Molecular Dynamics Simulation with the Experiment
Liquids 2023, 3(3), 314-370; https://doi.org/10.3390/liquids3030021 - 16 Aug 2023
Abstract
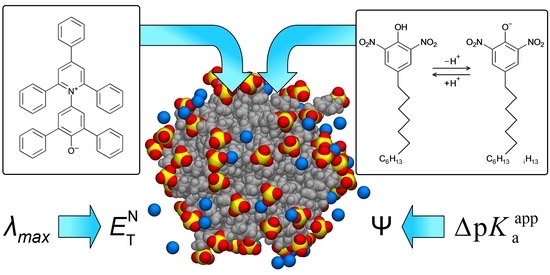
This article summarizes a series of seventeen publications by the authors devoted to molecular dynamics modeling of various indicator dyes (molecular probes) enclosed in surfactant micelles. These dyes serve as generally recognized tools for studying various types of organized solutions, among which surfactant
[...] Read more.
This article summarizes a series of seventeen publications by the authors devoted to molecular dynamics modeling of various indicator dyes (molecular probes) enclosed in surfactant micelles. These dyes serve as generally recognized tools for studying various types of organized solutions, among which surfactant micelles in water are the simplest and most explored. The modeling procedure involves altogether 50 to 95 surfactant molecules, 16 to 28 thousand water molecules, and a single dye molecule. The presentation of the simulation results was preceded by a brief review of the state of experimental studies. This article consists of three parts. First, despite numerous literature data devoted to modeling the micelles itself, we decided to revisit this issue. The structure and hydration of the surface of micelles of surfactants, first of all of sodium n-dodecylsulfate, SDS, and cetyltrimethylammonium bromide, CTAB, were studied. The values of the electrical potential, Ψ, were estimated as functions of the ionic strength and distance from the surface. The decrease in the Ψ value with distance is gradual. Attempts to consider both DS− and CTA+ micelles in water without counterions result in a decay into two smaller aggregates. Obviously, the hydrophobic interaction (association) of the hydrocarbon tails balances the repulsion of the charged headgroups of these small “bare” micelles. The second part is devoted to the study of seven pyridinium N-phenolates, known as Reichardt’s dyes, in ionic micelles. These most powerful solvatochromic indicators are now used for examining various colloidal systems. The localization and orientation of both zwitterionic and (colorless) cationic forms are generally consistent with intuitive ideas about the hydrophobicity of substituents. Hydration has been quantitatively described for both the dye molecule as a whole and the oxygen atom. A number of markers, including the visible absorption spectra of Reichardt’s dyes, enable assuming a better hydration of the micellar surface of SDS than that of CTAB. However, our data show that it is more correct to speak about the more pronounced hydrogen-bonding ability of water molecules in anionic micelles than about better hydration of the SDS micelles as compared to CTAB ones. Finally, a set of acid–base indicators firmly fixed in the micellar pseudophase were studied by molecular dynamics. They are instruments for estimating electrostatic potentials of micelles and related aggregates as Ψ
(This article belongs to the Special Issue Solvatochromic Probes and Their Applications in Molecular Interaction Studies—a Themed Issue to Honor Professor Dr. Christian Reichardt)
►▼
Show Figures
Graphical abstract
Open AccessArticle
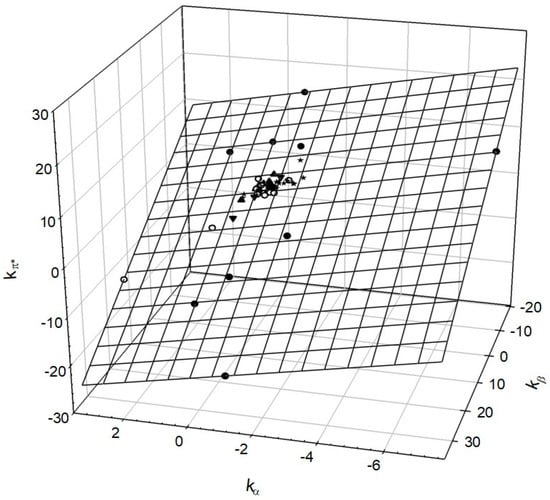
Reichardt’s Dye-Based Solvent Polarity and Abraham Solvent Parameters: Examining Correlations and Predictive Modeling
Liquids 2023, 3(3), 303-313; https://doi.org/10.3390/liquids3030020 - 02 Aug 2023
Cited by 1
Abstract
The concept of “solvent polarity” is widely used to explain the effects of using different solvents in various scientific applications. However, a consensus regarding its definition and quantitative measure is still lacking, hindering progress in solvent-based research. This study hopes to add to
[...] Read more.
The concept of “solvent polarity” is widely used to explain the effects of using different solvents in various scientific applications. However, a consensus regarding its definition and quantitative measure is still lacking, hindering progress in solvent-based research. This study hopes to add to the conversation by presenting the development of two linear regression models for solvent polarity, based on Reichardt’s ET(30) solvent polarity scale, using Abraham solvent parameters and a transformer-based model for predicting solvent polarity directly from molecular structure. The first linear model incorporates the standard Abraham solvent descriptors s, a, b, and the extended model ionic descriptors j+ and j−, achieving impressive test-set statistics of R2 = 0.940 (coefficient of determination), MAE = 0.037 (mean absolute error), and RMSE = 0.050 (Root-Mean-Square Error). The second model, covering a more extensive chemical space but only using the descriptors s, a, and b, achieves test-set statistics of R2 = 0.842, MAE = 0.085, and RMSE = 0.104. The transformer-based model, applicable to any solvent with an associated SMILES string, achieves test-set statistics of R2 = 0.824, MAE = 0.066, and RMSE = 0.095. Our findings highlight the significance of Abraham solvent parameters, especially the dipolarity/polarizability, hydrogen-bond acidity/basicity, and ionic descriptors, in predicting solvent polarity. These models offer valuable insights for researchers interested in Reichardt’s ET(30) solvent polarity parameter and solvent polarity in general.
Full article
(This article belongs to the Special Issue Solvatochromic Probes and Their Applications in Molecular Interaction Studies—a Themed Issue to Honor Professor Dr. Christian Reichardt)
►▼
Show Figures
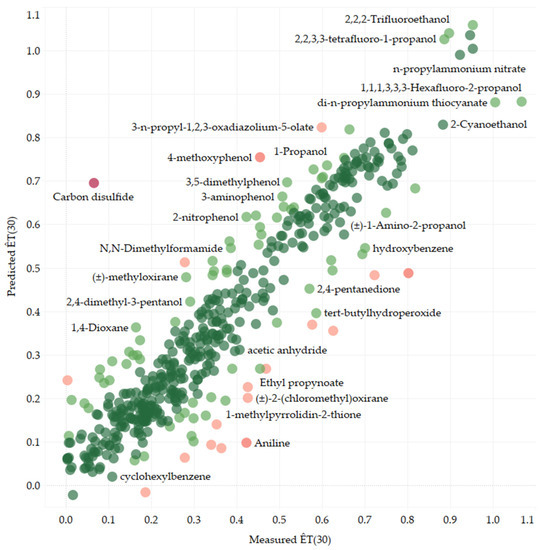
Figure 1
Open AccessArticle
An X-ray and Neutron Scattering Study of Aqueous MgCl2 Solution in the Gigapascal Pressure Range
by
, , , , and
Liquids 2023, 3(3), 288-302; https://doi.org/10.3390/liquids3030019 - 04 Jul 2023
Cited by 2
Abstract
The structure of electrolyte solutions under pressure at a molecular level is a crucial issue in the fundamental science of understanding the nature of ion solvation and association and application fields, such as geological processes on the Earth, pressure-induced protein denaturation, and supercritical
[...] Read more.
The structure of electrolyte solutions under pressure at a molecular level is a crucial issue in the fundamental science of understanding the nature of ion solvation and association and application fields, such as geological processes on the Earth, pressure-induced protein denaturation, and supercritical water technology. We report the structure of an aqueous 2 m (=mol kg−1) MgCl2 solution at pressures from 0.1 MPa to 4 GPa and temperatures from 300 to 500 K revealed by X-ray- and neutron-scattering measurements. The scattering data are analyzed by empirical potential structure refinement (EPSR) modeling to derive the pair distribution functions, coordination number distributions, angle distributions, and spatial density functions (3D structure) as a function of pressure and temperature. Mg2+ forms rigid solvation shells extended to the third shell; the first solvation shell of six-fold octahedral coordination with about six water molecules at 0 GPa transforms into about five water molecules and one Cl− due to the formation of the contact ion pairs in the GPa pressure range. The Cl− solvation shows a substantial pressure dependence; the coordination number of a water oxygen atom around Cl− increases from 8 at 0.1 MPa/300 K to 10 at 4 GPa/500 K. The solvent water transforms the tetrahedral network structure at 0.1 MPa/300 K to a densely packed structure in the GPa pressure range; the number of water oxygen atoms around a central water molecule gradually increases from 4.6 at 0.1 MPa/298 K to 8.4 at 4 GPa/500 K.
Full article
(This article belongs to the Special Issue Hydration of Ions in Aqueous Solution)
►▼
Show Figures
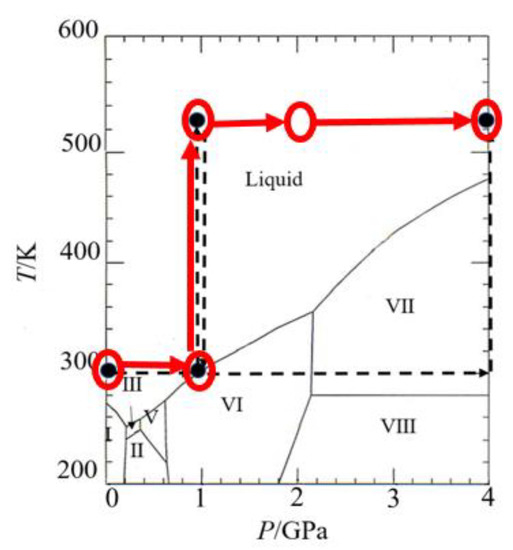
Figure 1
Open AccessArticle
Hydration of Phosphate Ion in Polarizable Water: Effect of Temperature and Concentration
by
and
Liquids 2023, 3(3), 278-287; https://doi.org/10.3390/liquids3030018 - 21 Jun 2023
Abstract
The hydration of phosphate ions, an essential component of many biological molecules, is studied using all-atom molecular dynamics (MD) simulation and quantum chemical methods. MD simulations are carried out by employing a mean-field polarizable water model. A good linear correlation between the self-diffusion
[...] Read more.
The hydration of phosphate ions, an essential component of many biological molecules, is studied using all-atom molecular dynamics (MD) simulation and quantum chemical methods. MD simulations are carried out by employing a mean-field polarizable water model. A good linear correlation between the self-diffusion coefficient and phosphate anion concentration is ascertained from the computed mean-square displacement (MSD) profiles. The HB dynamics of the hydration of the phosphate anion is evaluated from the time-dependent autocorrelation function CHB(t) and is determined to be slightly faster for the phosphate–anion system as compared to that of the water–water system at room temperature. The coordination number (CN) of the phosphate ion is found to be 15.9 at 298 K with 0.05 M phosphate ion concentration. The average CN is also calculated to be 15.6 for the same system by employing non-equilibrium MD simulation, namely, the well-tempered meta-dynamics method. A full geometry optimization of the PO43−·16H2O cluster is investigated at the ωB97X-D/aug-cc-pVTZ level of theory, and the hydration of the phosphate anion is observed to have both singly and doubly bonded anion–water hydrogen bonds and inter-water hydrogen bonds in a range between 0.169–0.201 nm and 0.192–0.215 nm, respectively. Modified Stokes–Einstein relation is used to calculate the conductivity of the phosphate ion and is found to be in good agreement with the experimentally observed value.
Full article
(This article belongs to the Special Issue Hydration of Ions in Aqueous Solution)
►▼
Show Figures
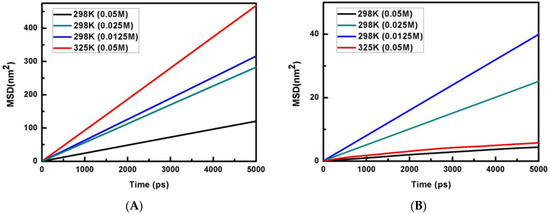
Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Crystals, Liquids, Materials, Molecules, Nanomaterials
Recent Advances in Liquid Crystals
Topic Editors: Jiatong Sun, Xiaoqian WangDeadline: 31 March 2024

Conferences
Special Issues
Special Issue in
Liquids
Solubility and Solubilization of Drugs: Modeling and Thermodynamic Analysis
Guest Editor: Daniel Ricardo DelgadoDeadline: 31 January 2024
Special Issue in
Liquids
Recent Advances in the Behavior of Liquids in Honor of Prof. Dr. William Acree Jr.
Guest Editors: William E. Acree, Jr., Juan SaavedraDeadline: 31 May 2024
Special Issue in
Liquids
Energy Transfer in Liquids
Guest Editor: Darin J. UlnessDeadline: 31 July 2024
Special Issue in
Liquids
Nanocarbon-Liquid Systems
Guest Editor: Nikolay O. Mchedlov-PetrossyanDeadline: 31 August 2024
Topical Collections
Topical Collection in
Liquids
Feature Papers in Solutions and Liquid Mixtures Research
Collection Editors: Enrico Bodo, Federico Marini