-
 Tributyl(1-((dimethylamino)(dimethyliminio)methyl)-1,4-dihydropyridin-4-yl)phosphonium Ditrifluoromethanesulfonate
Tributyl(1-((dimethylamino)(dimethyliminio)methyl)-1,4-dihydropyridin-4-yl)phosphonium Ditrifluoromethanesulfonate -
![Synthesis and Characterization of 2-(((2,7-Dihydroxynaphthalen-1-yl)methylene)amino)-3′,6′-bis(ethylamino)-2′,7′-dimethylspiro[isoindoline-1,9′-xanthen]-3-one and Colorimetric Detection of Uranium in Water](https://pub.mdpi-res.com/title_story/title_story_17011716204987.jpg?1705329949) Synthesis and Characterization of 2-(((2,7-Dihydroxynaphthalen-1-yl)methylene)amino)-3′,6′-bis(ethylamino)-2′,7′-dimethylspiro[isoindoline-1,9′-xanthen]-3-one and Colorimetric Detection of Uranium in Water
Synthesis and Characterization of 2-(((2,7-Dihydroxynaphthalen-1-yl)methylene)amino)-3′,6′-bis(ethylamino)-2′,7′-dimethylspiro[isoindoline-1,9′-xanthen]-3-one and Colorimetric Detection of Uranium in Water -
![2-(2-Fluoro-[1,1′-biphenyl]-4-yl)-<em>N</em>-(4-methyl-2-oxo-2<em>H</em>-chromen-7-yl)propanamide](https://pub.mdpi-res.com/title_story/title_story_17010855127956.jpg?1705329949) 2-(2-Fluoro-[1,1′-biphenyl]-4-yl)-N-(4-methyl-2-oxo-2H-chromen-7-yl)propanamide
2-(2-Fluoro-[1,1′-biphenyl]-4-yl)-N-(4-methyl-2-oxo-2H-chromen-7-yl)propanamide -
 Methyl-4-hydroxy-2-(2-hydroxypropan-2-yl)-6-methyl-2,3-dihydrobenzofuran-5-carboxylate
Methyl-4-hydroxy-2-(2-hydroxypropan-2-yl)-6-methyl-2,3-dihydrobenzofuran-5-carboxylate -
 Cocrystal of Codeine and Cyclopentobarbital
Cocrystal of Codeine and Cyclopentobarbital
Journal Description
Molbank
Molbank
is an international, peer-reviewed, open access journal comprised of a unique collection of one-compound-per-paper short notes on synthetic compounds and natural products published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, ESCI (Web of Science), Reaxys, CAPlus / SciFinder, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 15.5 days after submission; acceptance to publication is undertaken in 2.6 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
0.6 (2022)
Latest Articles
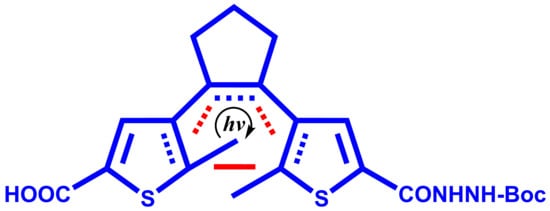
4-(2-(5-(2-(tert-Butoxycarbonyl)hydrazinecarbonyl)-2-methylthiophen-3-yl)cyclopent-1-enyl)-5-methylthiophene-2-carboxylic Acid
Molbank 2024, 2024(1), M1760; https://doi.org/10.3390/M1760 - 11 Jan 2024
Abstract
Diarylethene (DAE) molecular photoswitches draw attention as building units in the preparation of diverse photoactive molecules. An interesting class of these molecules are photoactive peptides. A way to build DAE moiety into peptides/peptidomimetics is via DAE amino acids, an example of which has
[...] Read more.
Diarylethene (DAE) molecular photoswitches draw attention as building units in the preparation of diverse photoactive molecules. An interesting class of these molecules are photoactive peptides. A way to build DAE moiety into peptides/peptidomimetics is via DAE amino acids, an example of which has been demonstrated in bioactive cyclic peptides, wherein the DAE Fmoc-amino acid was prepared and used. Herein, the preparation of DAE Boc-amino acid is presented using a modified method of synthesis. This contribution to the DAE amino acid collection could be useful in the further enhancement of diversity in designing different routes to photoactive peptides.
Full article
(This article belongs to the Section Organic Synthesis)
►
Show Figures
Open AccessShort Note
5,5′-Bis[9-(2-ethylhexyl)-9H-carbazol-3-yl]-4,4′-diphenyl-2,2′-bithiazole
Molbank 2024, 2024(1), M1761; https://doi.org/10.3390/M1761 - 11 Jan 2024
Abstract
Stille coupling between 5,5′-dibromo-4,4′-diphenyl-2,2′-bithiazole and 9-(2-ethylhexyl)-3-(tributylstannyl)-9H-carbazole in the presence of Pd(Ph3P)2Cl2 in toluene, heated at reflux for 2 h, gave 5,5′-bis[9-(2-ethylhexyl)-9H-carbazol-3-yl]-4,4′-diphenyl-2,2′-bithiazole in 85% yield.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures

Figure 1
Open AccessShort Note
9-Ethyl-6,6-dimethyl-8-[4-(morpholin-4-yl)piperidin-1-yl]-11-oxo-6,11-dihydro-5H-benzo[b]carbazole-3-carbonitrile Hydrochloride
Molbank 2024, 2024(1), M1759; https://doi.org/10.3390/M1759 - 05 Jan 2024
Abstract
Alectinib hydrochloride is an anticancer medication used for the first-line treatment of non-small cell lung cancer. Although it was approved for medical use ten years ago, and three polymorphs of this substance were proposed based on X-ray diffraction patterns, their crystal structures remained
[...] Read more.
Alectinib hydrochloride is an anticancer medication used for the first-line treatment of non-small cell lung cancer. Although it was approved for medical use ten years ago, and three polymorphs of this substance were proposed based on X-ray diffraction patterns, their crystal structures remained unknown to date. The main problem was the preparation of high quality single crystals due to the very low solubility of the salt. Herein, we report on the molecular and crystal structure of form I of alectinib hydrochloride as obtained using powder X-ray diffraction data from a laboratory source. Short Cl…N distances between the anion and the nitrogen atoms of the morpholine and benzo[b]carbazole rings indicate the positions of the H(N) atoms. As a result, the cation and anion form infinite Cl…H(N)-bonded chains.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures

Figure 1
Open AccessCommunication
(5Z,9Z)-14-[(3,28-Dioxoolean-12-en-28-yl)oxy]tetradeca-5,9-dienoic Acid with Cytotoxic Activity
Molbank 2024, 2024(1), M1758; https://doi.org/10.3390/M1758 - 02 Jan 2024
Abstract
For the first time, a synthetic analogue of natural (5Z,9Z)-dienoic acid has been synthesized in the form of a hybrid molecule containing a fragment of oleanolic acid and (5Z,9Z)-tetradeca-5.9-dienedicarboxylic acid, synthesized using a new reaction of Ti-catalyzed homo-cyclomagnesiation 1,2-dienes. The high cytotoxic activity
[...] Read more.
For the first time, a synthetic analogue of natural (5Z,9Z)-dienoic acid has been synthesized in the form of a hybrid molecule containing a fragment of oleanolic acid and (5Z,9Z)-tetradeca-5.9-dienedicarboxylic acid, synthesized using a new reaction of Ti-catalyzed homo-cyclomagnesiation 1,2-dienes. The high cytotoxic activity of (5Z,9Z)-14-[(3,28-dioxoolean-12-en-28-yl)oxy]tetradeca-5,9-dienoic acid against tumor cells Jurkat, K562, U937 and HL60 was established. This compound is also an inducer of apoptosis, affects the cell cycle and inhibits human topoisomerase I.
Full article
(This article belongs to the Section Natural Products)
►▼
Show Figures

Figure 1
Open AccessShort Note
3-(4-Bromophenyl)-1-carbamothioyl-5-(2-carbamothioylhydrazinyl)-4,5-dihydro-1H-pyrazole-5-carboxylic Acid
Molbank 2024, 2024(1), M1757; https://doi.org/10.3390/M1757 - 02 Jan 2024
Abstract
The reaction of 4-(4-bromophenyl)-2,4-dioxobutanoic acid with thiosemicarbazide, in a ratio of 1:2, when boiled in ethanol gives 3-(4-bromophenyl)-1-carbamothioyl-5-(2-carbamothioylhydrazinyl)-4,5-dihydro-1H-pyrazole-5-carboxylic acid with a good yield. This compound was fully characterized.
Full article
(This article belongs to the Collection Heterocycle Reactions)
►▼
Show Figures
Figure 1
Open AccessShort Note
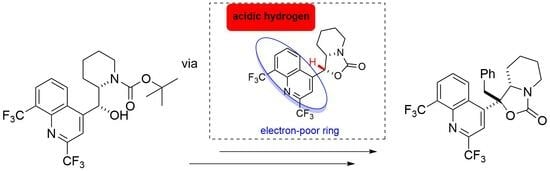
(1R/S,7aS/R)-1-Benzyl-1-[2,8-bis(trifluoromethyl)quinolin-4-yl]-hexahydro-oxazolo[3,4-a]pyridin-3-one
Molbank 2024, 2024(1), M1756; https://doi.org/10.3390/M1756 - 30 Dec 2023
Abstract
An unexpected diastereoselective C-alkylation of a mefloquine derivative in up to 57% yield was the result of an attempted Williamson etherification of Boc-mefloquine. The domino reaction involved oxazolidinone ring closure, deprotonation, and stereoselective carbon–carbon bond formation. The structure was confirmed with 2D NMR
[...] Read more.
An unexpected diastereoselective C-alkylation of a mefloquine derivative in up to 57% yield was the result of an attempted Williamson etherification of Boc-mefloquine. The domino reaction involved oxazolidinone ring closure, deprotonation, and stereoselective carbon–carbon bond formation. The structure was confirmed with 2D NMR experiments.
Full article
(This article belongs to the Collection Molecules from Side Reactions)
►▼
Show Figures
Graphical abstract
Open AccessCommunication
Unexpected Formation of the Iodobismuthate Salt (C14H15S2N2)2(C9H10SN)2[Bi4I16] upon Reaction of the Unsaturated Ligand Z-PySCH2CH=CHCH2SPy with BiI3
by
, , , , , and
Molbank 2024, 2024(1), M1755; https://doi.org/10.3390/M1755 - 29 Dec 2023
Abstract
The olefinic dithioether (Z)-1,4-bis(pyridin-2-ylthio)but-2-ene Z-PyS(CH2CH=CHCH2)SPy (L) was prepared by the treatment of cis-ClCH2CH=CHCH2Cl with in situ generated potassium pyridine-2-thiolate Py-SK and analyzed by IR and NMR spectroscopy. To investigate
[...] Read more.
The olefinic dithioether (Z)-1,4-bis(pyridin-2-ylthio)but-2-ene Z-PyS(CH2CH=CHCH2)SPy (L) was prepared by the treatment of cis-ClCH2CH=CHCH2Cl with in situ generated potassium pyridine-2-thiolate Py-SK and analyzed by IR and NMR spectroscopy. To investigate the chemistry of polynuclear iodobismuthate complexes, two equivalents of BiI3 were reacted with L in the MeOH solution to afford the anionic tetranuclear title compound (C14H15S2N2)2(C9H10SN)2[Bi4I116] with a N-protonated (Z)-1,4-bis(pyridin-2-ylthio)but-2-ene as a counterion. Compound 1 was characterized by IR and UV spectroscopy; the formation of a tetranuclear framework was ascertained by a single-crystal X-ray diffraction study performed at 100 K. Furthermore, an unusual Bi(III)-meditated cyclization of one Z-PyS(CH2CH=CHCH2)SPy ligand occurred, affording the bicyclic pyridinium salt 3-vinyl-2,3-dihydrothiazolo[3,2-a]pyridinium bearing a terminal vinyl group, compensating the second negative charge of the Bi4I164− cluster anion. The SCXRD characterization was completed by a Hirshfeld surface analysis, revealing some secondary interactions occurring in the crystal.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures

Figure 1
attachment
Supplementary material:
Supplementary File 1 (PDF, 1741 KiB)
Supplementary File 2 (MOL, 5 KiB)
Supplementary File 3 (INCHI, 2 KiB)
Supplementary File 4 (MOL, 5 KiB)
Supplementary File 5 (MOL, 5 KiB)
Supplementary File 6 (INCHI, 2 KiB)
Supplementary File 7 (MOL, 5 KiB)
Supplementary File 8 (MOL, 5 KiB)
Supplementary File 9 (INCHI, 2 KiB)
Supplementary File 10 (MOL, 5 KiB)
Supplementary File 1 (PDF, 1741 KiB)
Supplementary File 2 (MOL, 5 KiB)
Supplementary File 3 (INCHI, 2 KiB)
Supplementary File 4 (MOL, 5 KiB)
Supplementary File 5 (MOL, 5 KiB)
Supplementary File 6 (INCHI, 2 KiB)
Supplementary File 7 (MOL, 5 KiB)
Supplementary File 8 (MOL, 5 KiB)
Supplementary File 9 (INCHI, 2 KiB)
Supplementary File 10 (MOL, 5 KiB)
Open AccessCommunication
New Antimicrobial Accramycins from Streptomyces sp. MA37 Variant
Molbank 2024, 2024(1), M1754; https://doi.org/10.3390/M1754 - 28 Dec 2023
Abstract
In our continued desire to isolate more bioactive compounds from the Streptomyces sp. MA37 variant, ΔaccJ, three new accramycin derivatives have been successfully characterised. The structures of accramycin L-N (1–3) were established by high-resolution mass spectrometry and 1D and 2D nuclear
[...] Read more.
In our continued desire to isolate more bioactive compounds from the Streptomyces sp. MA37 variant, ΔaccJ, three new accramycin derivatives have been successfully characterised. The structures of accramycin L-N (1–3) were established by high-resolution mass spectrometry and 1D and 2D nuclear magnetic resonance. The antimicrobial evaluation of accramycin L-N against Staphylococcus aureus, Klebsiella pneumoniae, and Enterobacter cloacae showed minimum inhibitory concentration (MIC) values ranging from 0.77 to 13.02 µg/mL. Accramycin L exhibited the most significant activity against S. aureus. In addition, accramycin L-N (1–3) displayed significant activity against K. pneumoniae at the MIC values of 0.81, 0.77, and 0.79 µg/mL, respectively.
Full article
(This article belongs to the Section Natural Products)
►▼
Show Figures

Figure 1
Open AccessShort Note
N,N′-Dipropyloxamide
by
, , , , , and
Molbank 2024, 2024(1), M1753; https://doi.org/10.3390/M1753 - 22 Dec 2023
Abstract
N,N′-Dipropyloxamide (1) was synthesised by the reaction between diethyloxalate and n-propylamine in ethanol. Compound 1 was fully characterised by both microanalytical (elemental analysis, melting point determination) and spectroscopic means (FT-IR and NMR spectroscopy). Crystals suitable for single
[...] Read more.
N,N′-Dipropyloxamide (1) was synthesised by the reaction between diethyloxalate and n-propylamine in ethanol. Compound 1 was fully characterised by both microanalytical (elemental analysis, melting point determination) and spectroscopic means (FT-IR and NMR spectroscopy). Crystals suitable for single crystal X-ray diffraction were isolated by the slow evaporation of a methyl alcohol solution of the compound. The resulting crystal structure shows the prominent role exerted by intermolecular hydrogen bonds in the crystal packing.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures

Figure 1
Open AccessShort Note
Bis(2,6-di(pyridin-2-yl)pyridin-4-yl)-6,6′-(1,2-diselanediyl)dihexanoate
by
, , , and
Molbank 2024, 2024(1), M1752; https://doi.org/10.3390/M1752 - 20 Dec 2023
Abstract
►▼
Show Figures
The present paper describes the preparation and characterization of a new dinuclear ligand based on terpyridine featuring a diselenide unit. This new compound was synthesized in a two-step procedure that first involved the insertion of the diselenide moiety on a carboxylic acid and
[...] Read more.
The present paper describes the preparation and characterization of a new dinuclear ligand based on terpyridine featuring a diselenide unit. This new compound was synthesized in a two-step procedure that first involved the insertion of the diselenide moiety on a carboxylic acid and was followed by a Steglich esterification reaction between the biscarboxylic acid containing the diselenide unit and 2,6-di(pyridin-2-yl)pyridin-4-ol (tpyOH). The title compound was characterized via FT-IR, Raman, NMR (1D and 2D), and UV-Vis spectroscopies and elemental analysis. Emission properties were investigated.
Full article

Figure 1
Open AccessShort Note
(Z)-1-Benzyl-5-(4-bromophenyl)-5-hydroxy-4-(2-oxomorpholin-3-ylidene)pyrrolidine-2,3-dione
Molbank 2023, 2023(4), M1751; https://doi.org/10.3390/M1751 - 18 Dec 2023
Abstract
The reaction of 8-(4-bromobenzoyl)-3,4-dihydro-1H-pyrrolo[2,1-c][1,4]oxazine-1,6,7-trione with benzylamine in acetonitrile at room temperature afforded a good yield of (Z)-1-benzyl-5-(4-bromophenyl)-5-hydroxy-4-(2-oxomorpholin-3-ylidene)pyrrolidine-2,3-dione. The compound was fully characterized.
Full article
(This article belongs to the Collection Heterocycle Reactions)
►▼
Show Figures
Figure 1
Open AccessShort Note
4-(5-Benzyl-3-((4-fluorophenyl)sulfonyl)-5-methyl-4,5-dihydrofuran-2-yl)-2-nitrobenzamide
by
, , , , and
Molbank 2023, 2023(4), M1750; https://doi.org/10.3390/M1750 - 15 Dec 2023
Abstract
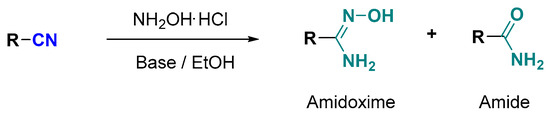
As part of our ongoing attempt to broaden the applications of the amidoxime moiety as a potential source of new antileishmanial agents, this study focuses on the product 4-(5-Benzyl-3-((4-fluorophenyl)sulfonyl)-5-methyl-4,5-dihydrofuran-2-yl)-2-nitrobenzamide. This unexpected amide was obtained in an 85% yield as the major product with
[...] Read more.
As part of our ongoing attempt to broaden the applications of the amidoxime moiety as a potential source of new antileishmanial agents, this study focuses on the product 4-(5-Benzyl-3-((4-fluorophenyl)sulfonyl)-5-methyl-4,5-dihydrofuran-2-yl)-2-nitrobenzamide. This unexpected amide was obtained in an 85% yield as the major product with a conventional amidoxime synthesis protocol (Ethanol/Na2CO3) involving the reaction of hydroxylamine and a nitrile group. The formation of this amide derivative instead of the expected amidoxime can be attributed to two complementary effects: the strong electron effect of the nitro group and the influence of ethanol, a polar protic solvent. Alternatively, the desired amidoxime derivative, 4-(5-benzyl-3-((4-fluorophenyl)sulfonyl)-5-methyl-4,5-dihydrofuran-2-yl)-N′-hydroxy-2-nitrobenzimidamide, was obtained in an 80% yield by an alternative protocol (DMSO/KOtBu). This original compound, featuring a nitro group in the ortho position to the amidoxime, will be further evaluated, both in the field of medicinal chemistry and in other relevant areas, highlighting an unusual method to access amidoximes from hindered substrates.
Full article
(This article belongs to the Collection Molecules from Side Reactions)
►▼
Show Figures
Scheme 1
Open AccessShort Note
3-Benzoyl-2-hydroxy-3a-[(3-methylquinoxalin-2-yl)methyl]-1H-pyrrolo[2,1-c][1,4]benzothiazine-1,4(3aH)-dione
Molbank 2023, 2023(4), M1749; https://doi.org/10.3390/M1749 - 13 Dec 2023
Abstract
The reaction of 3-benzoylpyrrolo[2,1-c][1,4]benzothiazine-1,2,4-trione with 2,3-dimethylquinoxaline afforded 3-benzoyl-2-hydroxy-3a-[(3-methylquinoxalin-2-yl)methyl]-1H-pyrrolo[2,1-c][1,4]benzothiazine-1,4(3aH)-dione in a moderate yield. The compound was fully characterized.
Full article
(This article belongs to the Collection Heterocycle Reactions)
►▼
Show Figures
Figure 1
attachment
Supplementary material:
Supplementary File 1 (PDF, 959 KiB)
Supplementary File 2 (MOL, 2 KiB)
Supplementary File 3 (INCHI, 739 B)
Supplementary File 4 (MOL, 2 KiB)
Supplementary File 5 (MOL, 15 KiB)
Supplementary File 6 (INCHI, 6 KiB)
Supplementary File 7 (MOL, 15 KiB)
Supplementary File 8 (MOL, 15 KiB)
Supplementary File 9 (INCHI, 6 KiB)
Supplementary File 10 (MOL, 15 KiB)
Supplementary File 11 (MOL, 16 KiB)
Supplementary File 12 (INCHI, 7 KiB)
Supplementary File 13 (MOL, 17 KiB)
Supplementary File 14 (ZIP, 2271 KiB)
Supplementary File 1 (PDF, 959 KiB)
Supplementary File 2 (MOL, 2 KiB)
Supplementary File 3 (INCHI, 739 B)
Supplementary File 4 (MOL, 2 KiB)
Supplementary File 5 (MOL, 15 KiB)
Supplementary File 6 (INCHI, 6 KiB)
Supplementary File 7 (MOL, 15 KiB)
Supplementary File 8 (MOL, 15 KiB)
Supplementary File 9 (INCHI, 6 KiB)
Supplementary File 10 (MOL, 15 KiB)
Supplementary File 11 (MOL, 16 KiB)
Supplementary File 12 (INCHI, 7 KiB)
Supplementary File 13 (MOL, 17 KiB)
Supplementary File 14 (ZIP, 2271 KiB)
Open AccessCommunication
Building Triazolated Macrocycles from Bis-Propargylated Calix[4]arenes and Bis-Azidomethylated Azobenzene or Stilbene
by
, , , , , and
Molbank 2023, 2023(4), M1748; https://doi.org/10.3390/M1748 - 07 Dec 2023
Abstract
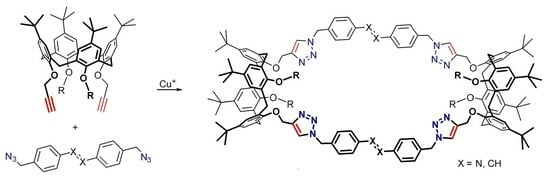
Copper(I)-catalyzed azide-alkyne cycloaddition was employed to construct biscalixarene assemblies from the calix[4]arene dipropargyl ethers and 4,4′-bis-azidomethylated azobenzene or stilbene. Three bis(calixarenes) having the calix[4]arene cores linked to each other by pairs of (E)-azobenzene/stilbene units through four triazole groups were obtained as
[...] Read more.
Copper(I)-catalyzed azide-alkyne cycloaddition was employed to construct biscalixarene assemblies from the calix[4]arene dipropargyl ethers and 4,4′-bis-azidomethylated azobenzene or stilbene. Three bis(calixarenes) having the calix[4]arene cores linked to each other by pairs of (E)-azobenzene/stilbene units through four triazole groups were obtained as confirmed by NMR, HRMS and X-ray diffraction data. Nevertheless, the formation of larger macrocycles and polymeric/oligomeric products was found to be the major competing process that seriously limited the applicability of the one-step macrocyclization approach for the construction of photoresponsive biscalixarene assemblies linked by pairs of azobenzene/stilbene units.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures
Graphical abstract
attachment
Supplementary material:
Supplementary File 1 (PDF, 389 KiB)
Supplementary File 2 (MOL, 2 KiB)
Supplementary File 3 (INCHI, 794 B)
Supplementary File 4 (MOL, 2 KiB)
Supplementary File 5 (MOL, 2 KiB)
Supplementary File 6 (INCHI, 849 B)
Supplementary File 7 (MOL, 2 KiB)
Supplementary File 8 (MOL, 2 KiB)
Supplementary File 9 (INCHI, 779 B)
Supplementary File 10 (MOL, 2 KiB)
Supplementary File 11 (MOL, 2 KiB)
Supplementary File 12 (INCHI, 848 B)
Supplementary File 13 (MOL, 2 KiB)
Supplementary File 14 (ZIP, 115 KiB)
Supplementary File 1 (PDF, 389 KiB)
Supplementary File 2 (MOL, 2 KiB)
Supplementary File 3 (INCHI, 794 B)
Supplementary File 4 (MOL, 2 KiB)
Supplementary File 5 (MOL, 2 KiB)
Supplementary File 6 (INCHI, 849 B)
Supplementary File 7 (MOL, 2 KiB)
Supplementary File 8 (MOL, 2 KiB)
Supplementary File 9 (INCHI, 779 B)
Supplementary File 10 (MOL, 2 KiB)
Supplementary File 11 (MOL, 2 KiB)
Supplementary File 12 (INCHI, 848 B)
Supplementary File 13 (MOL, 2 KiB)
Supplementary File 14 (ZIP, 115 KiB)
Open AccessCommunication
5,8-Quinolinedione Attached to Quinone Derivatives: XRD Diffraction, Fourier Transform Infrared Spectra and Computational Analysis
by
, , , , and
Molbank 2023, 2023(4), M1747; https://doi.org/10.3390/M1747 - 28 Nov 2023
Abstract
Quinoline and isoquinoline moieties occur in many natural and synthetic compounds exhibiting high biological activity. The purpose of this study was to analyze the chemical structures of 5,8-quinolinedione and 5,8-isoquinoline derivatives using FT-IR spectroscopy supplemented with theoretical DFT calculations. Spectroscopic measurements were conducted
[...] Read more.
Quinoline and isoquinoline moieties occur in many natural and synthetic compounds exhibiting high biological activity. The purpose of this study was to analyze the chemical structures of 5,8-quinolinedione and 5,8-isoquinoline derivatives using FT-IR spectroscopy supplemented with theoretical DFT calculations. Spectroscopic measurements were conducted using the attenuated total reflection (ATR) mode in the frequency range of 4000–400 cm−1. An analysis of FT-IR spectra was carried out, assigning the characteristic vibration frequencies of various functional groups to individual peaks. It was found that the experimental and calculated FT-IR spectra showed a good correlation for all the compounds under study. The most significant difference in the spectra occurred in the region of carbonyl bands. For compounds with the 5,8-quinolinedione moiety, two separated C=O vibration peaks were observed, while for compounds with the 5,8-isoquinolinedione moiety, the carbonyl vibrations created only one peak. This difference makes it possible to distinguish between the 5,8-quinolinedione and 5,8-isoquinolinedione derivatives.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures

Figure 1
attachment
Supplementary material:
Supplementary File 1 (PDF, 2364 KiB)
Supplementary File 2 (MOL, 3 KiB)
Supplementary File 3 (INCHI, 874 B)
Supplementary File 4 (MOL, 3 KiB)
Supplementary File 5 (MOL, 3 KiB)
Supplementary File 6 (INCHI, 914 B)
Supplementary File 7 (MOL, 3 KiB)
Supplementary File 8 (MOL, 3 KiB)
Supplementary File 9 (INCHI, 962 B)
Supplementary File 10 (MOL, 3 KiB)
Supplementary File 11 (MOL, 3 KiB)
Supplementary File 12 (INCHI, 971 B)
Supplementary File 13 (MOL, 3 KiB)
Supplementary File 14 (MOL, 3 KiB)
Supplementary File 15 (INCHI, 1 KiB)
Supplementary File 16 (MOL, 3 KiB)
Supplementary File 17 (MOL, 3 KiB)
Supplementary File 18 (INCHI, 1 KiB)
Supplementary File 19 (MOL, 3 KiB)
Supplementary File 20 (MOL, 3 KiB)
Supplementary File 21 (INCHI, 1 KiB)
Supplementary File 22 (MOL, 3 KiB)
Supplementary File 23 (MOL, 4 KiB)
Supplementary File 24 (INCHI, 1 KiB)
Supplementary File 25 (MOL, 3 KiB)
Supplementary File 1 (PDF, 2364 KiB)
Supplementary File 2 (MOL, 3 KiB)
Supplementary File 3 (INCHI, 874 B)
Supplementary File 4 (MOL, 3 KiB)
Supplementary File 5 (MOL, 3 KiB)
Supplementary File 6 (INCHI, 914 B)
Supplementary File 7 (MOL, 3 KiB)
Supplementary File 8 (MOL, 3 KiB)
Supplementary File 9 (INCHI, 962 B)
Supplementary File 10 (MOL, 3 KiB)
Supplementary File 11 (MOL, 3 KiB)
Supplementary File 12 (INCHI, 971 B)
Supplementary File 13 (MOL, 3 KiB)
Supplementary File 14 (MOL, 3 KiB)
Supplementary File 15 (INCHI, 1 KiB)
Supplementary File 16 (MOL, 3 KiB)
Supplementary File 17 (MOL, 3 KiB)
Supplementary File 18 (INCHI, 1 KiB)
Supplementary File 19 (MOL, 3 KiB)
Supplementary File 20 (MOL, 3 KiB)
Supplementary File 21 (INCHI, 1 KiB)
Supplementary File 22 (MOL, 3 KiB)
Supplementary File 23 (MOL, 4 KiB)
Supplementary File 24 (INCHI, 1 KiB)
Supplementary File 25 (MOL, 3 KiB)
Open AccessCommunication
Synthesis and In Silico Drug-Likeness Modeling of 5-FU/ASA Hybrids
Molbank 2023, 2023(4), M1745; https://doi.org/10.3390/M1745 - 27 Nov 2023
Abstract
A series of 5-FU-ASA hybrids were synthesized with good yields using click chemistry as the key step. The structures of these compounds were elucidated by spectroscopic analysis. Finally, an optimal pharmacokinetic profile was also estimated for each synthetized hybrid. Taken together, hybrids 4a
[...] Read more.
A series of 5-FU-ASA hybrids were synthesized with good yields using click chemistry as the key step. The structures of these compounds were elucidated by spectroscopic analysis. Finally, an optimal pharmacokinetic profile was also estimated for each synthetized hybrid. Taken together, hybrids 4a–h could be used as starting points for further pharmacological studies concerning therapeutic cancer intervention.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures

Figure 1
Open AccessShort Note
1-(2,5-Dimethoxy-4-nitrophenyl)piperidine
Molbank 2023, 2023(4), M1744; https://doi.org/10.3390/M1744 - 27 Nov 2023
Abstract
►▼
Show Figures
Treatment of the non-purified mixture of dinitro isomers obtained from the nitration of 1,4-dimethoxybenzene with piperidine led to the isolation of novel but minor adduct, 1-(2,5-dimethoxy-4-nitrophenyl)piperidine (2b) in 15% yield. Yields of nucleophilic aromatic substitution adducts are high when using purified
[...] Read more.
Treatment of the non-purified mixture of dinitro isomers obtained from the nitration of 1,4-dimethoxybenzene with piperidine led to the isolation of novel but minor adduct, 1-(2,5-dimethoxy-4-nitrophenyl)piperidine (2b) in 15% yield. Yields of nucleophilic aromatic substitution adducts are high when using purified 1,4-dimethoxy-2,5-dinitrobenzene (1b) with piperidine and pyrrolidine to give (2b) and 1-(2,5-dimethoxy-4-nitrophenyl)pyrrolidine (3b) in 76% and 82% yield, respectively.
Full article

Graphical abstract
Open AccessShort Note
2-(4-(Fluorosulfonyloxy)phenyl)benzoxazole
by
, , , and
Molbank 2023, 2023(4), M1746; https://doi.org/10.3390/M1746 - 27 Nov 2023
Abstract
New 2-(4-(fluorosulfonyloxy)phenyl)benzoxazole (2) was synthesized through the SuFEx click reaction in a two-chamber reactor. The effect of silylation on the yield of the target compound was investigated. The fluorescent properties of compound 2 were determined using experimental and computational methods.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures

Figure 1
attachment
Supplementary material:
Supplementary File 1 (PDF, 782 KiB)
Supplementary File 2 (MOL, 3 KiB)
Supplementary File 3 (INCHI, 1 KiB)
Supplementary File 4 (MOL, 3 KiB)
Supplementary File 5 (MOL, 2 KiB)
Supplementary File 6 (INCHI, 610 B)
Supplementary File 7 (MOL, 2 KiB)
Supplementary File 8 (MOL, 3 KiB)
Supplementary File 9 (INCHI, 1 KiB)
Supplementary File 10 (MOL, 3 KiB)
Supplementary File 11 (MOL, 3 KiB)
Supplementary File 12 (INCHI, 1 KiB)
Supplementary File 13 (MOL, 3 KiB)
Supplementary File 1 (PDF, 782 KiB)
Supplementary File 2 (MOL, 3 KiB)
Supplementary File 3 (INCHI, 1 KiB)
Supplementary File 4 (MOL, 3 KiB)
Supplementary File 5 (MOL, 2 KiB)
Supplementary File 6 (INCHI, 610 B)
Supplementary File 7 (MOL, 2 KiB)
Supplementary File 8 (MOL, 3 KiB)
Supplementary File 9 (INCHI, 1 KiB)
Supplementary File 10 (MOL, 3 KiB)
Supplementary File 11 (MOL, 3 KiB)
Supplementary File 12 (INCHI, 1 KiB)
Supplementary File 13 (MOL, 3 KiB)
Open AccessCommunication
Synthesis of Norabietyl and Nordehydroabietyl Imidazolidine-2,4,5-Triones and Their Activity against Tyrosyl-DNA Phosphodiesterase 1
by
, , , , , , , and
Molbank 2023, 2023(4), M1743; https://doi.org/10.3390/M1743 - 09 Nov 2023
Abstract
New imidazolidine-2,4,5-triones with norabietic, nordehydroabietic, and adamantane substituents were synthesized by reacting oxalyl chloride and the corresponding ureas, providing good yields. Bioisosteric replacement of the ureide group with a parabanic acid fragment made it possible to increase the solubility of compounds and conduct
[...] Read more.
New imidazolidine-2,4,5-triones with norabietic, nordehydroabietic, and adamantane substituents were synthesized by reacting oxalyl chloride and the corresponding ureas, providing good yields. Bioisosteric replacement of the ureide group with a parabanic acid fragment made it possible to increase the solubility of compounds and conduct biological studies. The compounds inhibit the DNA repair enzyme tyrosyl-DNA phosphodiesterase 1 in submicromolar concentrations. Cytotoxic concentrations were also studied on the glioblastoma cell line SNB19.
Full article
(This article belongs to the Section Natural Products)
►▼
Show Figures

Figure 1
Open AccessShort Note
4,4′,4″-(Benzene-1,3,5-triyltris(ethyne-2,1-diyl))tris(1-methylpyridin-1-ium) Iodide
Molbank 2023, 2023(4), M1742; https://doi.org/10.3390/M1742 - 08 Nov 2023
Abstract
►▼
Show Figures
Despite having been known for a long time, quaternary 4,4′-bipyridinium salts, or viologens, are still a highly inspiring class of compounds, thanks to their peculiar redox and charge transfer properties. However, more complex structures containing multiple pyridinium rings, also interspaced by conjugated moieties,
[...] Read more.
Despite having been known for a long time, quaternary 4,4′-bipyridinium salts, or viologens, are still a highly inspiring class of compounds, thanks to their peculiar redox and charge transfer properties. However, more complex structures containing multiple pyridinium rings, also interspaced by conjugated moieties, allow an even wider synthetic variability and tunability of their characteristics. The compound described herein is a star-shaped, fully conjugated molecule with three methylated pyridinium rings connected by a triple bond spacer to a central benzene core, which was synthesized from readily available building blocks, representing a quite simple model of multi-pyridyl extended viologen; its UV–visible absorption and fluorescence spectra have also been investigated.
Full article

Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Catalysts, Chemistry, Materials, Molbank, Molecules
Catalysis: Homogeneous and Heterogeneous, 2nd Edition
Topic Editors: Luísa Margarida Martins, Isidro M. PastorDeadline: 31 October 2024
Topic in
Catalysts, Chemistry, Inorganics, Molbank, Molecules, Polymers
Heterocyclic Carbene Catalysis
Topic Editors: Sabine Berteina-Raboin, Thierry Besson, Patrick RollinDeadline: 30 November 2024
Topic in
Catalysts, Chemistry, Molbank, Molecules, Sustainable Chemistry
Towards the Sustainable Synthesis of Biologically Active Molecules in Green Solvents
Topic Editors: Antonio Salomone, Serena PerroneDeadline: 31 December 2024

Conferences
Special Issues
Topical Collections
Topical Collection in
Molbank
Molecules from Catalytic Processes
Collection Editor: Nicola Della Ca’